Is trypsin found in the stomach
 Rachel Young
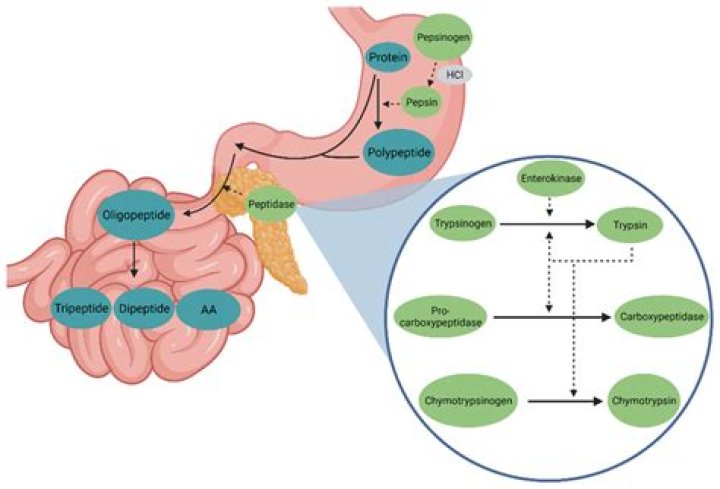
Rachel Young In the small intestine, trypsin breaks down proteins, continuing the process of digestion that began in the stomach. It may also be referred to as a proteolytic enzyme, or proteinase. Trypsin is produced by the pancreas in an inactive form called trypsinogen.
Is pepsin or trypsin found in the stomach?
Origin: Pepsin is the chief digestive enzyme in stomach, which is produced by the gastric gland in stomach and is a component of gastric juice, while trypsin in produced by the pancreas and is a component of pancreatic juice.
Why does trypsin not work in the stomach?
Once it reaches small intestine, it is converted in active trypsin by enterokinase enzyme. Trypsin acts in alkaline medium and it is prevented from taking any damaging action on intestinal lining of lumen due to presence of protective mucus layer on it.
What enzyme is found in the stomach?
Pepsin is a stomach enzyme that serves to digest proteins found in ingested food. Gastric chief cells secrete pepsin as an inactive zymogen called pepsinogen. Parietal cells within the stomach lining secrete hydrochloric acid that lowers the pH of the stomach.Where is trypsin and chymotrypsin found?
Two of the main enzymes involved in this process are trypsin and chymotrypsin. Both of these enzymes come from the pancreas and act in the duodenum (the portion right between the stomach and small intestines, or the first section of the small intestines).
What is the difference between trypsin and lipase?
Trypsin and lipase are both digestive enzymes with rather dissimilar properties and structure. Trypsin is a protease and catalyzes hydrolysis of proteins while lipase catalyzes hydrolysis of lipids.
Where is pepsin found?
An enzyme made in the stomach that breaks down proteins in food during digestion. Stomach acid changes a protein called pepsinogen into pepsin.
What is the means of trypsin?
Definition of trypsin : a proteolytic enzyme that is secreted in the pancreatic juice in the form of trypsinogen, is activated in the duodenum, and is most active in a slightly alkaline medium.What does trypsin digest?
Trypsin is an enzyme that helps us digest protein. In the small intestine, trypsin breaks down proteins, continuing the process of digestion that began in the stomach. It may also be referred to as a proteolytic enzyme, or proteinase. Trypsin is produced by the pancreas in an inactive form called trypsinogen.
What are the 4 main digestive enzymes?- Amylase.
- Maltase.
- Lactase.
- Lipase.
- Proteases.
- Sucrase.
Where trypsin is found?
Trypsin is a proteolytic enzyme found in the lumen of the small intestine and widely expressed in other tissues.
Where can you find trypsin?
Trypsin (EC 3.4. 21.4) is a serine protease from the PA clan superfamily, found in the digestive system of many vertebrates, where it hydrolyzes proteins. Trypsin is formed in the small intestine when its proenzyme form, the trypsinogen produced by the pancreas, is activated.
What is the product of trypsin?
The products of trypsin digestion are amino acids and various polypeptides.
How does trypsin digest casein?
Trypsin works in the small intestine, after acid and pepsin in the stomach have commenced the work of breaking down the proteins. This experiment uses milk which contains the protein casein. As the casein in milk break down, the smaller molecules become soluble, thereby reducing the opacity of the fluid.
Where is chymotrypsin found?
Chymotrypsin is an enzyme that digests protein in the small intestine. This test measures the amount of chymotrypsin in stool to help evaluate whether your pancreas is functioning properly. Chymotrypsinogen, the inactive precursor of chymotrypsin, is produced in the pancreas and transported to the small intestine.
Where are trypsin chymotrypsin and carboxypeptidase produced?
The exocrine pancreas secretes three endopeptidases (trypsin, chymotrypsin, and elastase) and two exopeptidases (carboxypeptidase A and carboxypeptidase B) in inactive forms.
What is trypsin and pepsin?
Pepsin is an enzyme which acts only in acidic medium ,trypsine is an enzyme which acts in alkaline medium. … Pepsin is secreted in stomach, trypsin is secreted in small intestine. Pepsin is situated in gastric glands, trypsin is situated in pancreas.
Where is salivary amylase found?
Salivary amylase is a glucose-polymer cleavage enzyme that is produced by the salivary glands. It comprises a small portion of the total amylase excreted, which is mostly made by the pancreas.
Where is pancreatic lipase found?
Pancreatic lipase is secreted into the duodenum through the duct system of the pancreas. Its concentration in serum is normally very low.
Where is lipase and trypsin secreted?
The pancreas secretes digestive enzymes into the duodenum and hormones into the bloodstream. The digestive enzymes (such as amylase, lipase, and trypsin) are released from the cells of the acini and flow into the pancreatic duct.
What is the function of lipase and trypsin?
Amylase digests carbohydrates, lipase digests fats, and trypsin digests proteins. The pancreas also secretes large amounts of sodium bicarbonate, which protects the duodenum by neutralizing the acid that comes from the stomach.
What produces lipase in the body?
Lipase is an enzyme the body uses to break down fats in food so they can be absorbed in the intestines. Lipase is produced in the pancreas, mouth, and stomach.
Can trypsin digest itself?
“Without efforts to stabilize it, trypsin will eventually digest itself,” says Tracy Adair-Kirk, Principal Scientist at MilliporeSigma. This is particularly undesirable for applications where autolysis may contaminate and confound experimental results.
Why does trypsin digest protein?
Trypsin cleaves the peptide bond between the carboxyl group of arginine or the carboxyl group of lysine and the amino group of the adjacent amino acid. The rate of cleavage occurs more slowly when the lysine and arginine residues are adjacent to acidic amino acids in the sequence or cystine.
How many amino acids are in trypsin?
The active site of trypsin is composed of three amino acids, which are serine 195, histidine 57, and aspartate 102, called a catalytic triad.
How was trypsin discovered?
Trypsin was first discovered in 1876 by Kuhne, who investigated the proteolytic activity of the enzyme. In 1931 the enzyme was purified by crystallization by Norothrop and Kunitz and later in 1974 the three dimensional structure of trypsin was determined. … Anionic trypsin or Trypsin-2 is expressed in the pancreas.
What is trypsin in cell culture?
Trypsinization is the process of cell dissociation using trypsin, a proteolytic enzyme which breaks down proteins, to dissociate adherent cells from the vessel in which they are being cultured. When added to a cell culture, trypsin breaks down the proteins which enable the cells to adhere to the vessel.
Where does trypsin cleave proteins?
Trypsin cleaves peptides on the C-terminal side of lysine and arginine amino acid residues. If a proline residue is on the carboxyl side of the cleavage site, the cleavage will not occur. If an acidic residue is on either side of the cleavage site, the rate of hydrolysis has been shown to be slower.
What type of enzymes do lysosomes contain?
Lysosomes are membrane-enclosed compartments filled with hydrolytic enzymes that are used for the controlled intracellular digestion of macromolecules. They contain about 40 types of hydrolytic enzymes, including proteases, nucleases, glycosidases, lipases, phospholipases, phosphatases, and sulfatases.
What are the 5 enzymes?
- Amylase, produced in the mouth. …
- Pepsin, produced in the stomach. …
- Trypsin, produced in the pancreas. …
- Pancreatic lipase, produced in the pancreas. …
- Deoxyribonuclease and ribonuclease, produced in the pancreas.
Is Epi a real thing?
Exocrine pancreatic insufficiency (EPI) is a condition characterized by deficiency of the exocrine pancreatic enzymes, resulting in the inability to digest food properly, or maldigestion.