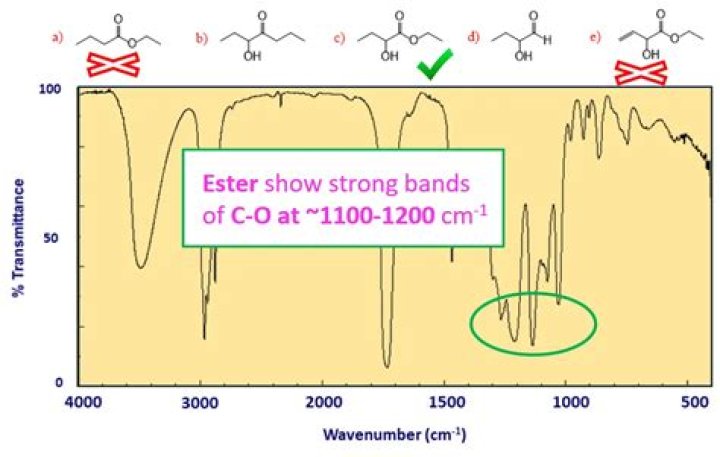
Is Jasmine an ester
 Emma Terry
Emma Terry Benzyl acetate is an organic ester with the molecular formula C9H10O2. … Natural sources of benzyl acetate include varieties of flowers like jasmine (Jasminum), and fruits like pear, apple, etc.
What is an ester example?
Examples of Esters Ethyl acetate (ethyl ethanoate) is an ester. … Other examples of esters include ethyl propanoate, propyl methanoate, propyl ethanoate, and methyl butanoate. Glycerides are fatty acid esters of glycerol.
What esters are in perfumes?
5240302010
What is an ester group?
Esters are a functional group commonly encountered in organic chemistry. They are characterized by a carbon bound to three other atoms: a single bond to a carbon, a double bond to an oxygen, and a single bond to an oxygen. … Ester names are derived from the parent alcohol and the parent acid.What is ester formula?
Esters have the general formula RCOOR′, where R may be a hydrogen atom, an alkyl group, or an aryl group, and R′ may be an alkyl group or an aryl group but not a hydrogen atom. … The esters shown here are ethyl acetate (a) and methyl butyrate (b). Esters occur widely in nature.
What products contain esters?
Esters are responsible for the aroma of many fruits, including apples, durians, pears, bananas, pineapples, and strawberries. Several billion kilograms of polyesters are produced industrially annually, important products being polyethylene terephthalate, acrylate esters, and cellulose acetate.
What is the simplest ester?
Methyl formate, also called methyl methanoate, is the methyl ester of formic acid. The simplest example of an ester, it is a colorless liquid with an ethereal odour, high vapor pressure, and low surface tension. It is a precursor to many other compounds of commercial interest.
What two functional groups make an ester?
Esters. Esters are functional groups produced from the condensation of an alcohol with a carboxylic acid, and are named based on these components.Which two compounds can form ester?
Esters may also be obtained by reaction of acid halides or acid anhydrides with alcohols or by reaction of salts of carboxylic acids with alkyl halides. One ester may be converted to another ester by reaction (transesterified) with an alcohol, a carboxylic acid, or a third ester in the presence of a catalyst.
Is ester a carbonyl group?Both carboxylic acids and esters contain a carbonyl group with a second oxygen atom bonded to the carbon atom in the carbonyl group by a single bond. In a carboxylic acid, the second oxygen atom also bonds to a hydrogen atom.
Article first time published onAre esters sweet smelling?
– The ester formed by the acetic acid with ethanol is sweet in smell. – The intermolecular force of attraction between the esters is weak. – Due to this less intermolecular force of attraction the ester compounds are volatile in nature. … – This volatile nature of esters makes us smell.
What ester smells like butterscotch?
The range of esters adds the fruitiness to rum’s aroma; particularly important contributors are ethyl propanoate which contributes a caramel-like, fruity aroma, and ethyl isobutyrate which has a butterscotch-like aroma.
How do you make an ester?
Esters are produced when carboxylic acids are heated with alcohols in the presence of an acid catalyst. The catalyst is usually concentrated sulphuric acid. Dry hydrogen chloride gas is used in some cases, but these tend to involve aromatic esters (ones where the carboxylic acid contains a benzene ring).
How do you name an ester?
Esters are named as if the alkyl chain from the alcohol is a substituent. No number is assigned to this alkyl chain. This is followed by the name of the parent chain from the carboxylic acid part of the ester with an –e remove and replaced with the ending –oate.
Is aspirin an ester?
Aspirin is a trade name for acetylsalicylic acid, a common analgesic. Acetylsalicylic acid is an acetic acid ester derivative of salicylic acid.
Is formate an ester?
Formate (IUPAC name: methanoate) is the anion derived A formate (compound) is a salt or ester of formic acid.
Is methyl formate an ester?
Methyl formate, a clear, volatile liquid, is the simplest carboxylate ester. It can be made in the lab by the acid-catalyzed esterification of formic acid and methanol.
Is methyl acetate an ester?
Methyl acetate, also known as MeOAc, acetic acid methyl ester or methyl ethanoate, is a carboxylate ester with the formula CH3COOCH3. It is a flammable liquid with a characteristically pleasant smell reminiscent of some glues and nail polish removers.
What are natural esters?
Natural esters are made from renewable natural sources, for example MIDEL eN 1204 (rapeseed/canola) and MIDEL eN 1215 (soybean). The base oil is chosen to give the best possible fit to the application; however unlike synthetic esters the properties of these base oils cannot be significantly altered.
What are esters in skin care?
Esters are fatty acids formed when an organic acid combines with alcohol or glycerin; they are natural and come with a whole host of benefits. They can enhance the feel and performance of other ingredients helping smooth the skin’s texture and acting as protection. They can also be great surfactants.
Where are esters found?
Esters are ubiquitous. Most naturally occurring fats and oils are the fatty acid esters of glycerol. Esters with low molecular weight are commonly used as fragrances and found in essential oils and pheromones.
Is ethyl acetate an ester?
Ethyl acetate is one of the simplest carboxylate esters. … Ethyl acetate is a widely used solvent, especially for paints, varnishes, lacquers, cleaning mixtures, and perfumes. Like last week’s MOTW, dichloromethane, it is used as a solvent for decaffeinating coffee beans.
Is Amine a functional group?
In organic chemistry, amines (/əˈmiːn, ˈæmiːn/, UK also /ˈeɪmiːn/) are compounds and functional groups that contain a basic nitrogen atom with a lone pair.
What is the difference between an ester and an ether?
The main difference between ether and ester lies in their structure. An ester group requires two oxygen atoms and two carbon atoms to complete its characteristic structure, while an ether group only needs one oxygen atom and two carbon atoms for its structure.
Which of the following is ester group?
The ester functional group is represented by –COOR. The suffix used for the functional ester is –oate. The given functional group is carboxylic acid in which carbon atom forms four bonds as it is a tetravalent compound.
What are esters how are they formed?
Esters are sweet smelling compounds. They are formed by the reaction of a carboxylic acid with an alcohol in the presence of conc. sulphuric acid. For example, ethyl ethanoate is prepared by reacting ethanoic acid and ethanol in the presence of conc.sulphuric acid.
Which of the following is called ester functional group?
Chemical classGroupFormulaCarbonateCarbonate esterROCOOR’CarboxylateCarboxylateRCOO−Carboxylic acidCarboxylRCOOHEsterCarboalkoxyRCOOR’
Is ether a carbonyl group?
Ethers are compounds with an oxygen atom bonded to two alkyl groups. Aldehydes and ketones contain the carbonyl functional group. In an aldehyde, the carbonyl is at the end of a carbon chain, while in a ketone, it is in the middle.
Why is ester not a carbonyl group?
The carbonyl group, a carbon-oxygen double bond, is the key structure in these classes of organic molecules: Aldehydes contain at least one hydrogen atom attached to the carbonyl carbon atom, ketones contain two carbon groups attached to the carbonyl carbon atom, carboxylic acids contain a hydroxyl group attached to …
What are ethers in chemistry?
ether, any of a class of organic compounds characterized by an oxygen atom bonded to two alkyl or aryl groups. … In an alcohol one hydrogen atom of a water molecule is replaced by an alkyl group, whereas in an ether both hydrogen atoms are replaced by alkyl or aryl groups.
What ester smells like bananas?
NamesChemical formulaC7H14O2Molar mass130.187 g·mol−1AppearanceColorless liquidOdorBanana-like