Is diarrhea isotonic or hypotonic
 Rachel Young
Rachel Young This is the type of dehydration most frequently caused by diarrhoea. It occurs when the net losses of water and sodium are in the same proportion as normally found in the ECF.
Is diarrhea hypertonic or hypotonic?
The most common causes of hypertonic dehydration are diarrhea, high fever, and vomiting. These can lead to dehydration and a salt-fluid imbalance.
Is vomiting isotonic or hypotonic?
characterised by isotonic loss of both water and solutes from the extracellular fluid (ECF) e.g., – vomiting, diarrhoea or through inadequate intake. no osmotic water shift from the intracellular fluid (ICF) to the ECF.
Is fluid loss in diarrhea hypotonic or isotonic?
In isotonic dehydration, there is an equal loss of water and sodium, and vomiting and diarrhea are the two most common causes. Hypotonic dehydration is caused by a greater loss of sodium than water. The use of diuretics is the most common cause of hypotonic dehydration.What causes hypotonic diarrhea?
Hypotonic dehydration occurs when sodium loss is greater than water loss, resulting in a decrease in serum osmolality. This causes a shift of water from the extracellular space into the intracellular space.
Does Diarrhoea cause isotonic dehydration?
Isotonic dehydration is often caused by diarrhea, vomiting or inadequate intake of fluid. Most commonly seen in infants, hypernatremic dehydration describes a loss of water that is greater than the amount of sodium lost, leading to a rise in blood sodium or hypernatremia.
Why is diarrhea isotonic?
Isotonic dehydration This is the type of dehydration most frequently caused by diarrhoea. It occurs when the net losses of water and sodium are in the same proportion as normally found in the ECF.
What is isotonic fluid loss?
There are several forms of dehydration. Isotonic water loss occurs when water and sodium are lost together. Causes of isotonic water loss are vomiting, diarrhea, sweating, burns, intrinsic kidney disease, hyperglycemia, and hypoaldosteronism. Hypertonic dehydration occurs when water losses exceed sodium losses.Is diarrhea insensible water loss?
Insensible losses refer to other routes of fluid loss, such as in sweat and from the respiratory tract. Fluid replacement goes beyond the normal physiologic losses and includes such conditions as vomiting, diarrhea, or severe cutaneous burns.
Why is a solution isotonic?The isotonic solution allow the cells to move water and nutrients in and out of the cells. This is necessary for blood cells to perform their function of delivering oxygen and other nutrients to other parts of the body.
Article first time published onWhy does diarrhea cause hypernatremia?
Hypovolemic hyponatremia can result from GI or renal losses of fluid. It was previously noted that vomiting and diarrhea typically involves fluid loss which has sodium + potassium concentration less than that of plasma and may result in hypernatremia.
Is water isotonic?
Isotonic solutions have the same water concentration on both sides of the cell membrane. Blood is isotonic. … Tapwater and pure water are hypotonic. A single animal cell ( like a red blood cell) placed in a hypotonic solution will fill up with water and then burst.
Is 10 dextrose hypertonic or hypotonic?
Dextrose 10% in Water (D10W) is an hypertonic IV solution used in the treatment of ketosis of starvation and provides calories (380 kcal/L), free water, and no electrolytes.
Why is isotonic used to treat dehydration?
Isotonic IV solutions restore fluid volume because they fill the tissues and maintain fluid volume more effectively than hypertonic or hypotonic solutions.
How is isotonic dehydration defined?
Isotonic dehydration is loss of water with preserved normal effective osmolality of body fluids. Causes include loss of isotonic fluids via the gastrointestinal tract, kidneys, or skin (burns); loss of blood; or as a result of fluid sequestration in the “third space” (eg, the peritoneum).
When would you use isotonic hypertonic and hypotonic solutions?
If a cell is placed in a hypertonic solution, water will leave the cell, and the cell will shrink. In an isotonic environment, there is no net water movement, so there is no change in the size of the cell. When a cell is placed in a hypotonic environment, water will enter the cell, and the cell will swell.
Does diarrhea cause acidosis or alkalosis?
Diarrhea is the most common cause of external loss of alkali resulting in metabolic acidosis. Biliary, pancreatic, and duodenal secretions are alkaline and are capable of neutralizing the acidity of gastric secretions.
Does diarrhea cause hyperkalemia?
The occurrence of diarrhea in combination with hyperkalemia is rather unusual. In most instances, watery diarrhea leads to K+ losses and hypokalemia.
What is the difference between osmotic and secretory diarrhea?
Osmotic diarrhea occurs when you eat substances that can’t be absorbed. Secretory diarrhea occurs when your body secretes electrolytes into your intestine. This causes water to build up.
Which electrolyte is lost in diarrhea?
Diarrhea can cause dehydration (when your body loses large amounts of water), electrolyte imbalance (loss of sodium, potassium and magnesium that play a key role in vital bodily functions) and kidney failure (not enough blood/fluid is supplied to the kidneys).
How do you balance electrolytes after diarrhea?
Drinking fruit juices and sports drinks, as well as eating soups with clear broths, can help replenish electrolyte levels. Just pay attention to the amount of sugar in these drinks, as too much sugar can worsen diarrhea symptoms.
Why is dextrose contraindicated in Diarrhoea?
Do not use 5% glucose (dextrose) solution or 0.18% saline with 5% dextrose solution, as they increase the risk for hyponatraemia, which can cause cerebral oedema.
Which are examples of preformed water?
- Water in an apple.
- Water in a baked potato and steak dinner.
- Water in a glass of milk.
What is insensible water loss quizlet?
Insensible water loss. Water loss that is not obvious like from lungs, skin and respiratory tract. Only $35.99/year. hypokalemia. Abnormally low potassium concentration in the blood resulting from decreased potassium intake and absorption or increased output from things like diarrhea, vomiting, and diuretics.
Which is an insensible mechanism of fluid loss?
Some fluid is lost through perspiration (part of the body’s temperature control mechanism) and as water vapor in exhaled air. These are termed “insensible fluid losses” as they cannot be easily measured.
Is sweat isotonic or hypertonic?
Primary sweat is nearly isotonic with blood plasma (e.g. approximately 135–145 mmol/L Na+, approximately 95–110 mmol/L Cl−, and approximately 4–5 mmol/L K+) [29, 46–49].
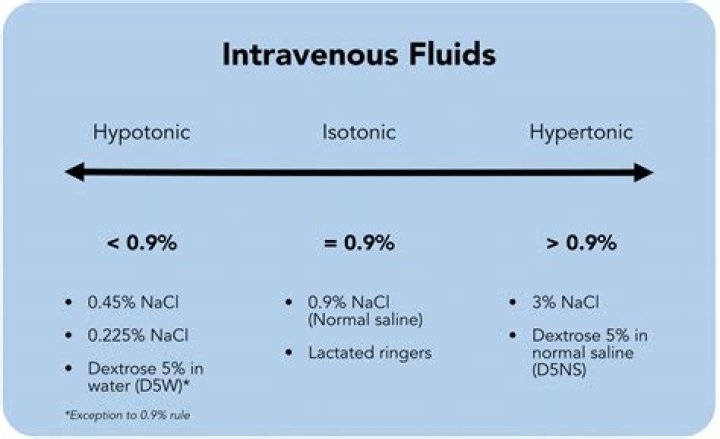
What are hypotonic solutions?
Hypotonic solution: A solution that contains fewer dissolved particles (such as salt and other electrolytes) than is found in normal cells and blood. Hypotonic solutions are commonly used to give fluids intravenously to hospitalized patients in order to treat or avoid dehydration.
What is hypertonic hypotonic and isotonic?
Isotonic: The solutions being compared have equal concentration of solutes. Hypertonic: The solution with the higher concentration of solutes. Hypotonic: The solution with the lower concentration of solutes.
What is an example of isotonic?
Common examples of isotonic solutions are 0.9% normal saline and lactated ringers. These fluids are useful when the patient has lost fluid volume from blood loss, trauma, or dehydration due to excessive nausea/vomiting or diarrhea.
What are examples of isotonic fluids?
Examples of isotonic solutions include normal saline (0.9% sodium chloride), lactated Ringer’s solution, 5% dextrose in water (D5W), and Ringer’s solution.
What is an example of a isotonic solution?
Isotonic solutions are IV fluids that have a similar concentration of dissolved particles as blood. An example of an isotonic IV solution is 0.9% Normal Saline (0.9% NaCl).