Copper(II) fluoride
| Names |
| Density | 4.23 g/cm3 (anhydrous) 2.934 g/cm3 (dihydrate) |
| Melting point | 836 °C (1,537 °F; 1,109 K) (anhydrous) 130 °C (dihydrate, decomposes) |
| Boiling point | 1,676 °C (3,049 °F; 1,949 K) (anhydrous) |
| Solubility in other solvents | Hygroscopic |
.
Besides, is cuf2 soluble?
Copper Fluoride is slightly soluble in water and has uses in ceramics and in fluxes used for brazing and soldering.
One may also ask, is cof2 soluble in water? CoF2 is sparingly soluble in water. The compound can be dissolved in warm mineral acid, and will decompose in boiling water.
Also, is CUBR soluble in water?
Copper(I) bromide
| Names |
| Solubility in water | slightly soluble |
| Solubility | soluble in HCl, HBr, ammonium hydroxide, acetonitrile negligible in acetone, sulfuric acid |
| Magnetic susceptibility (χ) | -49.0·10−6 cm3/mol |
| Refractive index (nD) | 2.116 |
Is KF aqueous?
Potassium fluoride. Description: Potassium fluoride appears as white powder or crystals with a sharp saline taste. Shipped as a solid or an aqueous solution.
Related Question Answers
Is CuF2 a Colour?
Out of Ag2SO4, CuF2 , MgF2 and CuCl, which compound will be coloured and why? As you can see there is one unpaired electron. This electron imparts colour to the compound. If all the electrons are paired up, the compound will not be coloured.What is the name for CuF2?
Molecular Formula: CuF2. Synonyms: Copper(II) fluoride. CuF2.Why do ionic compounds not need prefixes?
Do NOT use prefixes to indicate how many of each element is present; this information is implied in the name of the compound. since iron can form more than one charge. Ionic Compounds Containing a Metal and a Polyatomic Ion.What is the general rule for the solubility of nitrate salts?
Salts containing nitrate ion (NO3-) are generally soluble. Salts containing Cl -, Br -, or I - are generally soluble. Important exceptions to this rule are halide salts of Ag+, Pb2+, and (Hg2)2+. Thus, AgCl, PbBr2, and Hg2Cl2 are insoluble.What is the ionic formula for lithium iodide?
Lithium iodide | LiI - PubChem.What is the formula for chromium ii hydroxide?
Chromium(II) hydroxide | CrH2O2 - PubChem.What is the solubility of copper?
Copper was soluble at all concentrations in pH 5.5 distilled water. At higher pH values, the amount of soluble copper was a function of both pH and the total copper concentration, with a maximum of 4 mg/l soluble copper at pH 6.5 and a maximum of 1.3 mg/l soluble copper at pH 7.4.What happens to fluorine when it reacts with copper?
Fluorine reacts with copper to form an ionic compound. Each F gains an electron from the copper atom. This forms an F- ion with a full outer shell. The copper atom becomes a + ion.Is nh4br soluble in water?
Ammonium bromide, NH4Br, is the ammonium salt of hydrobromic acid. The chemical crystallizes in colorless prisms, possessing a saline taste; it sublimes on heating and is easily soluble in water.Is AgCl soluble in water?
Many ionic solids, such as silver chloride (AgCl) do not dissolve in water. The forces holding the solid AgCl lattice together are too strong to be overcome by the forces favoring the formation of the hydrated ions, Ag+(aq) and Cl-(aq).Is CuCl soluble in water?
Copper(I) chloride, commonly called cuprous chloride, is the lower chloride of copper, with the formula CuCl. The substance is a white solid sparingly soluble in water, but very soluble in concentrated hydrochloric acid.Is pbno32 soluble in water?
Lead(II) nitrate is an inorganic compound with the chemical formula Pb(NO3)2. It commonly occurs as a colourless crystal or white powder and, unlike most other lead(II) salts, is soluble in water.Is FeCO3 soluble in water?
Is FeCO3 Soluble in Water? FeCO3 is insoluble in water. Most ionic salts containing CO3 are insoluble in water with the exception of those involving ammonium or group one metallic elements.Is AgI soluble?
Answer and Explanation: AgI is practically insoluble in water. Only 3 x 10 -7 g will dissolve in 100mL of water at 20 °C. The solubility of most ionic compounds willIs CoF2 polar or nonpolar?
The electronegativity of fluorine is greater than chlorine, therefore the electron density is not symmetrical and the molecule is polar. The shape of SF6 S F 6 is Octahedral, and the electron density is symmetrical in nature so it is Non-Polar.Does KF dissolve in water?
KF is highly soluble in water. It initially dissociates into K+ and F-. However, the F- further hydrolyzes with water to form HF. Since HF is a weak acid (pKa = 3.17), it would rather exist as molecular HF than ionic F-.Is KF an acid or base?
KF is a salt that comes from a strong base, KOH, and a weak acid, HF. When added to water, the salt dissociates into a K+ ion and F- ion. The F- ion, which is the conjugate base of the weak acid, reacts with water by accepting one hydrogen atom from water and forming hydroxide.What is the value of KF for water?
Kf is the molal freezing point depression constant of the solvent (1.86 °C/m for water). m = molality = moles of solute per kilogram of solvent.Is KF polar or nonpolar?
Polar covalent bonds. Most real chemical bonds in nature are neither truly covalent nor truly ionic. Only homonuclear bonds are truly covalent, and nearly perfect ionic bonds can form between group I and group VII elements, for example, KF. 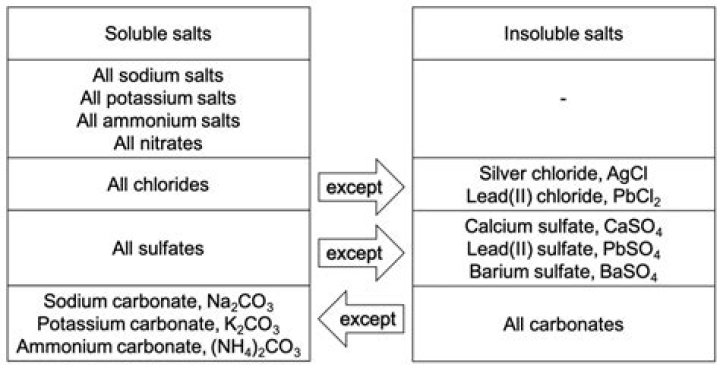
 William Burgess
William Burgess