Is common salt solution acidic or basic
 Andrew White
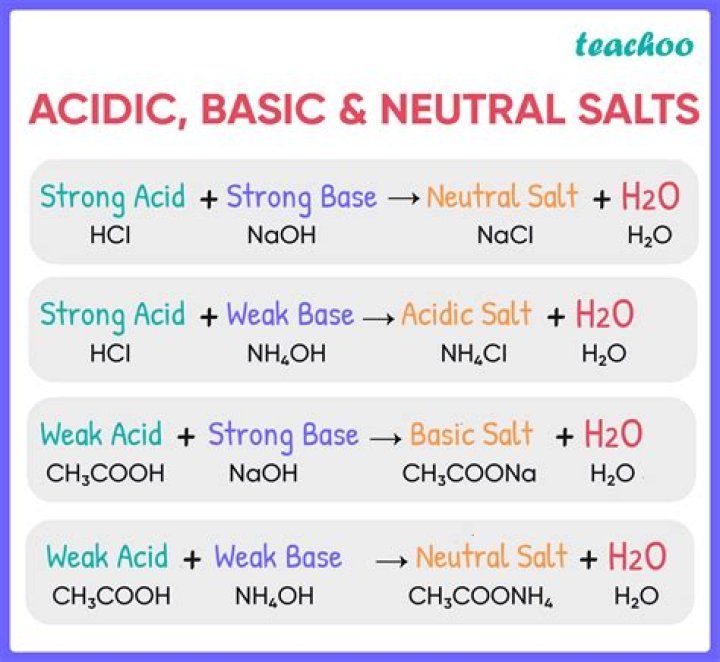
Andrew White The pH will remain neutral at 7. Halides and alkaline metals dissociate and do not affect the H+ as the cation does not alter the H+ and the anion does not attract the H+ from water. This is why NaCl is a neutral salt.
Is common salt an acid or base?
Keep in mind that a salt will only be basic if it contains the conjugate base of a weak acid. Sodium chloride, for instance, contains chloride (Cl–), which is the conjugate base of HCl. But because HCl is a strong acid, the Cl– ion is not basic in solution, and it isn’t capable of deprotonating water.
Why Common salt is neutral?
Salts that are from strong bases and strong acids do not hydrolyze. The pH will remain neutral at 7. Halides and alkaline metals dissociate and do not affect the H+ as the cation does not alter the H+ and the anion does not attract the H+ from water. This is why NaCl is a neutral salt.
Is common salt solution acidic?
The pH of salt is 7 which means that it is neutral in nature i.e. it is neither acidic nor basic.Is salt a base?
Is salt basic or acidic? The salt is basic only when it contains a weak acid conjugate base. For example, sodium chloride contains chloride (Cl-), the conjugate base of HCl.
Is common salt an acid or base explain your answer Class 7?
Answer: Sodium chloride is neutral in nature as it is a salt. It does not possess the properties of acids or bases.
Is common salt solution a basic?
Common salt is neutral in nature.
How do you know if a solution is acidic or basic?
If the pH is lower than 7, the solution is acidic. When pH is higher than 7, the solution is basic. These numbers describe the concentration of hydrogen ions in the solution and increase on a negative logarithmic scale.Which of the following salt solutions is acidic?
SaltAcidBaseSodium Carbonate(Na2CO3)HCO3(carbonic acid)NaOH(sodium hydroxide)Sodium Acetate(CH3COONa)CH3COOH(acetic acid)NaOHAmmonium Phosphate(NH4)3PO4H3PO4(Phosphoric acid)NH3(Ammonia)
What is salt base and acid?salt, in chemistry, substance produced by the reaction of an acid with a base. A salt consists of the positive ion (cation) of a base and the negative ion (anion) of an acid. The reaction between an acid and a base is called a neutralization reaction.
Article first time published onIs salt a mixture?
If we are talking about commercial grade salt (like table salt), you have a mixture. The salt is made up of multiple substances (sodium chloride, anti-caking agent, and supplements). These substances are not bonded to each other, and can be separated from each other.
Is common salt homogeneous or heterogeneous?
Homogeneous Mixtures The salt water described above is homogeneous because the dissolved salt is evenly distributed throughout the entire salt water sample. Often it is easy to confuse a homogeneous mixture with a pure substance because they are both uniform.
What is common salt Short answer?
Common salt is formed by the combination of hydrochloric acid and sodium hydroxide solution. It is the salt that we use in food. Sea water contains many salts dissolved in it. It is obtained on large scale from sea water by separating other salts from it. It may also be obtained from rock salt.
What is the pH value of common salt?
pH value of sodium chloride (common salt) is about equal to 7.
Which solution is basic in nature?
Ammonia turns red litmus to blue so it is basic in nature.
Which among the following salt solution is basic in nature?
Sodium acetate is basic in nature. It is a salt formed by neutralization of weak acid and strong base.
Which of the following salts solution is neutral *?
Detailed Solution The correct answer is NaCl. The neutral aqueous solution is a chemical solution which is neither acidic nor basic. The pH scale of this solution is close to 7. Salt is an ionic compound that is formed when an acid and a base neutralize each other.
Which of the following is solution is acidic in nature?
Apple Juice. ∴ Option A (Apple juice) is the correct answer.
What solutions are basic?
A basic solution is an aqueous solution containing more OH-ions than H+ions. In other words, it is an aqueous solution with a pH greater than 7. Basic solutions contain ions, conduct electricity, turn red litmus paper blue, and feel slippery to the touch.
Is milk acidic basic or neutral?
Milk — pasteurized, canned, or dry — is an acid-forming food. Its pH level is below neutral at about 6.7 to 6.9. This is because it contains lactic acid. Remember, though, that the exact pH level is less important than whether it’s acid-forming or alkaline-forming.
Which solution is the most acidic?
The most acidic solution is c. HF is a weak acid that partially ionizes. Despite having a higher concentration (1.0 M) than 0.50 M HCl, HCl will still have the lower pH as it possesses the higher concentration of hydrogen ions at equilibrium.
What makes a salt acidic?
Acid salts are a class of salts that produce an acidic solution after being dissolved in a solvent. Its formation as a substance has a greater electrical conductivity than that of the pure solvent. An acidic solution formed by acid salt is made during partial neutralization of diprotic or polyprotic acids.
Is carbonic acid acidic?
In aqueous solution carbonic acid behaves as a dibasic acid. The Bjerrum plot shows typical equilibrium concentrations, in solution, in seawater, of carbon dioxide and the various species derived from it, as a function of pH.
What do acids bases and salts have in common?
Common in acids and bases When acid and bases are dissolved in water solution, acid gives H+ ions whereas base gives OH– ions. So both acid and bases are electrolytes which conducts electricity.
Is common salt an element or compound or mixture?
Table salt is a compound consisting of equal parts of the elements sodium and chlorine. Salt cannot be separated into its two elements by filtering, distillation, or any other physical process.
Is salt a solution?
Salt dissolved in water is a solution. … For example, in a solution of salt in water, the solute is salt, and solvent is water. Solutions come in all phases, and the solvent and the solute do not have to be in the same phase to form a solution (such as salt and water).
Is common salt a compound or element?
Chemically, table salt consists of two elements, sodium (Na) and chloride (Cl). Neither element occurs separately and free in nature, but are found bound together as the compound sodium chloride.
Is salt a homogeneous solution?
How would a chemist categorize each example of matter? Saltwater acts as if it were a single substance even though it contains two substances—salt and water. Saltwater is a homogeneous mixture, or a solution. Soil is composed of small pieces of a variety of materials, so it is a heterogeneous mixture.
Is salt homogeneous?
Ordinary table salt is called sodium chloride. It is considered a substance because it has a uniform and definite composition. … The salt water described above is homogeneous because the dissolved salt is evenly distributed throughout the entire salt water sample.
Is salt a mixture or solution?
Salt water is a solution because it has these two characteristics: it has the same concentration of each of its parts throughout the solution, and it can be separated by some physical process.
When is common salt dissolved in water?
Water molecules pull the sodium and chloride ions apart, breaking the ionic bond that held them together. After the salt compounds are pulled apart, the sodium and chloride atoms are surrounded by water molecules. Once this happens, the salt is dissolved, resulting in a homogeneous solution.