Is Cobalt hydroxide a precipitate
 Christopher Martinez
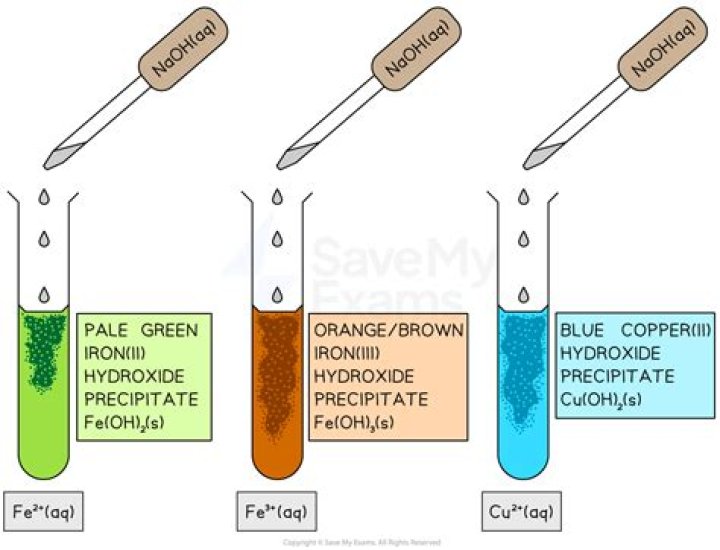
Christopher Martinez Cobalt(II) hydroxide precipitates as a solid when an alkali metal hydroxide is added to an aqueous solution of Co2+ salt.
Is cobalt III hydroxide soluble in water?
NamesOther names Cobaltic hydroxideIdentifiersCAS Number1307-86-43D model (JSmol)Interactive image
Is CoS soluble in water?
Compound FormulaCoSMelting Point1,195° CBoiling Point1425 °CDensity5.45 g/cm3Solubility in H2OInsoluble
Does cobalt chloride and sodium hydroxide form a precipitate?
Here, sodium hydroxide (NaOH) is added to cobalt(II) chloride (CoCl2). … The result is a red solution and a blue-green precipitate.Will cobalt II hydroxide precipitate in water?
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). ) is a pink solid insoluble in water.
Does cobalt chloride and sodium sulfate form a precipitate?
Here, sodium sulfide (Na2S) is added to cobalt(II) chloride (CoCl2). The result is a black precipitate.
How is cobalt hydroxide formed?
Cobalt hydroxide precipitate. Cobalt hydroxide precipitate (Co(OH)2) formed by adding 0.5 M solution of sodium hydroxide (NaOH) to a 0.2 M solution of cobalt chloride (CoCl2). The reaction is CoCl2 + KOH -> Co(OH)2 + KCl.
Is cobalt II nitrate soluble or insoluble?
Cobalt nitrate is the inorganic compound with the formula Co(NO3)2xH2O. It is cobalt(II)’s salt. The most common form is the hexahydrate Co(NO3)2·6H2O, which is a red-brown deliquescent salt that is soluble in water and other polar solvents.Is cobalt soluble?
Pure cobalt does not dissolve in water, but will dissolve (or react) with acids. … Compounds that are not soluble in water are cobalt carbonate and cobalt oxide. Compounds that are soluble in water are cobalt nitrate and cobalt sulfate.
Is carbonyl sulfide a dipole?NamesDipole moment0.65 DThermochemistryHeat capacity (C)41.5 J/mol KStd molar entropy (So298)231.5 J/mol K
Article first time published onWhat is cos chemistry?
Carbonyl sulfide (COS) is a colorless gas with a sulfur-like odor. Like carbon dioxide (CO2), its molecular structure is linear; but unlike CO2 it is flammable. It decomposes slowly in water and more rapidly in the presence of base.
Is cobalt sulfide a solid?
IdentifiersAppearanceblack solid (alpha) grayish-red crystals (beta)Density5.45 g/cm3Melting point1195 °CSolubility in water0.00038 g/100 mL (18 °C)
Is cobalt II hydroxide ionic?
Cobalt(II) hydroxide is a chemical compound. Its chemical formula is Co(OH)2. It contains cobalt in its +2 oxidation state as an ion. It also contains hydroxide ions.
Is NaOH insoluble in water?
When sodium hydroxide (NaOH) dissolves in water, it separates into positively – charged sodium ions (cations) and negatively – charged hydroxide ions (anions). … It is very soluble in water and, like all gases, it’s solubility depends on the partial pressure of the gas.
What is beryllium hydroxide formula?
Beryllium hydroxide has the formula Be(OH)2 and the molecular weight of 43.0268 g/mol. A more likely formula is Be(OH)+OH−. It can be prepared by reacting the oxide with water: BeO + H2O ⇒ Be(OH)2.
Is Cobalt hydroxide hygroscopic?
3.597Hygroscopic. Air sensitive. Incompatible with strong oxidizing agents.
Is cobalt an oxide?
Cobalt oxide is a family of chemical compounds consisting of cobalt and oxygen atoms. Compounds in the cobalt oxide family include: Cobalt(II) oxide (cobaltous oxide), CoO. Cobalt(III) oxide (cobaltic oxide), Co2O.
How do I know if a precipitate will form?
If the value of the ion product is greater than the value of the Ksp, then a precipitate will form. The formation of the precipitate lowers the concentration of each of the ions until the ion product is exactly equal to the Ksp, at which point precipitation ceases.
What creates a precipitate?
A precipitate is a solid formed in a chemical reaction that is different from either of the reactants. This can occur when solutions containing ionic compounds are mixed and an insoluble product is formed. … It also occurs in single displacement when one metal ion in solution is replaced by another metal ion.
What is precipitate and precipitation reaction?
A precipitation reaction refers to the formation of an insoluble salt when two solutions containing soluble salts are combined. The insoluble salt that falls out of solution is known as the precipitate, hence the reaction’s name. Precipitation reactions can help determine the presence of various ions in solution.
What is Co3 po42?
Cobalt(II) Phosphate. Alias: Cobaltous Phosphate. Formula: Co3(PO4)2.
Does Koh and fecl3 form a precipitate?
It is a precipitation reaction in which ferric and hydroxide ion on reaction with each other results in the formation of ferric hydroxide as reddish-brown precipitate. The other product is potassium chloride which remains in the solution.
When a solution of nitric acid and sodium hydroxide are mixed What precipitate if any is formed?
Explanation: Nitric acid and sodium hydroxide will neutralize each other in a 1:1 mole ratio, i.e. every mole of nitric acid will consume 1 mole of sodium hydroxide, to produce water and aqueous sodium nitrate, NaNO3 .
Does a reaction occur when aqueous solutions of cobalt II bromide and sodium hydroxide are combined?
There is no precipitate, insoluble gas, or water, so this reaction does not occur.
Does NaCl react with cocl2?
Here, cobalt(II) chloride (CoCl2) is added to sodium chloride (NaCl). … The result is no visible reaction.
Is cobalt sulfate a liquid?
Assay: 7.9-8.1% Cobalt. … Specific Gravity: 1.30 max. Appearance: Clear, dark red liquid.
Is cobalt solid liquid or gas?
Cobalt is a chemical element with symbol Co and atomic number 27. Classified as a transition metal, Cobalt is a solid at room temperature.
Is cobalt sulphate soluble in water?
NamesSolubility in wateranhydrous: 36.2 g/100 mL (20 °C) 38.3 g/100 mL (25 °C) 84 g/100 mL (100 °C) heptahydrate: 60.4 g/100 mL (3 °C) 67 g/100 mL (70 °C)
What type of bond is cobalt nitrate?
Cobalt nitrate is an Ionic compound because it is formed by the transferring of electrons that’s why it can be considered as an ionic compound….
Is cobalt II nitrate a cation or anion?
Cobalt dinitrate is a cobalt salt in which the cobalt metal is in the +2 oxidation state and the counter-anion is nitrate. It is an inorganic nitrate salt and a cobalt salt. It contains a cobalt(2+).
Is cobalt carbonate soluble?
NamesSolubility in waterCobalt (II) Carbonate is insoluble in distilled water.Solubility product (Ksp)1.0·10−10Solubilitysoluble in acid negligible in alcohol, methyl acetate insoluble in ethanolRefractive index (nD)1.855