Is ch4 a binary compound?
 William Burgess
William Burgess .
Furthermore, is methane a binary compound?
Binary compounds are materials/substances that exhibit only one type of strong chemical bond: metallic, ionic or covalent. But only methylene, CH2, and methane, CH4, possess only one type of strong chemical bond and are the only substances to be considered binaries, here.
Also, is NaCl a binary compound? In chemistry, a binary compound is something consisting of precisely two elements. In a binary compound, there may be only one of each element. We see this with sodium chloride (salt) NaCl, which has one sodium (Na) and one chlorine (Cl).
Also asked, is ch4 a compound or molecule?
All compounds are molecules but not all molecules are compounds. Molecular hydrogen (H2), molecular oxygen (O2) and molecular nitrogen (N2) are not compounds because each is composed of a single element. Water (H2O), carbon dioxide (CO2) and methane (CH4) are compounds because each is made from more than one element.
How do you know if a compound is binary?
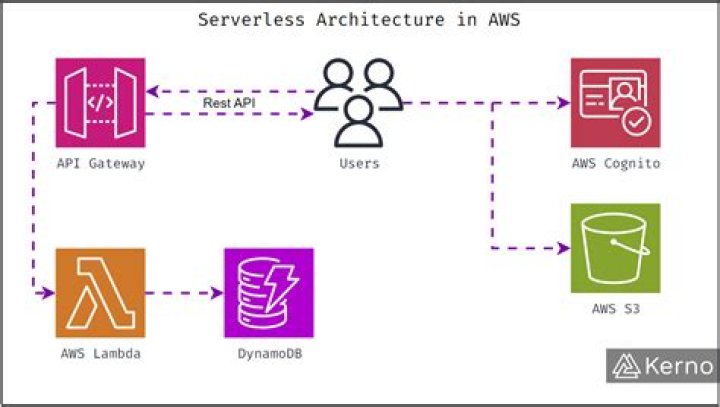
The order for names in a binary compound is first the cation, then the anion. Use the name of cation with a fixed oxidation state directly from the periodic table. The name of the anion will be made from the root of the element's name plus the suffix "-ide."
Related Question AnswersWhat is h2 called?
H2 is also called molecular hydrogen.It consists of two protons and two electrons. Consequently it is the most common form of Hydrogen because it is stable with a neutral charge. H2 is not a free radical. It is the antioxidant in 'hydrogen-rich' water. H2 is the smallest molecule in the universe.What are the two major types of binary compounds?
The major types of binary compounds are ionic (compounds that contain a metal and a nonmetal) and nonionic (compounds containing two nonmetals).What is a binary compound give three examples of binary compounds?
A binary compound contains only two elements. Examples are sodium chloride, water, and carbon dioxide.Can methane kill you?
In severe cases, there may be changes in breathing and heart rate, balance problems, numbness, and unconsciousness. If exposure is large or continues for a longer period it can kill. Skin or eye contact with liquefied methane released under pressure may cause frostbite. Can Methane cause cancer?What type of compound is ch4?
Methane (US: or UK: ) is a chemical compound with the chemical formula CH4 (one atom of carbon and four atoms of hydrogen). It is a group-14 hydride and the simplest alkane, and is the main constituent of natural gas.What does ch4 mean?
Methane (chemical formula CH4) is one of the simplest hydrocarbons, which literally means “containing hydrogen and carbon.”What produces the most methane?
But most sources of methane are of human origin — livestock and farming, decay in landfills, leakage from the oil and gas industry. Since 1750, the amount of methane in the atmosphere has doubled because of human activity. The oil and gas industry is the top contributor, creating one-third of all methane emissions.What is ch4 made up of?
Methane (US: /ˈm?θe?n/or UK: /ˈmiːθe?n/) is a chemical compound with the chemical formula CH4 (one atom of carbon and four atoms of hydrogen). It is a group-14 hydride and the simplest alkane, and is the main constituent of natural gas.Is h2s a molecular compound?
Hydrogen sulfide is the chemical compound with the formula H2S. It is a colorless chalcogen hydride gas with the characteristic foul odor of rotten eggs. H2S also occurs in volcanic gases, natural gas, and in some sources of well water. The human body produces small amounts of H2S and uses it as a signaling molecule.Is methane a stable molecule?
As the building block of all organic molecules, carbon is of particular interest to us. Carbon, with 4 electrons in its valence shell, will need another four electrons to fulfill the octet rule. Thus it needs to combine with 4 hydrogen atoms to form a stable compound called methane (CH4) as shown above.Is Co a diatomic molecule?
If a diatomic molecule consists of two atoms of the same element, such as hydrogen (H2) or oxygen (O2), then it is said to be homonuclear. Otherwise, if a diatomic molecule consists of two different atoms, such as carbon monoxide (CO) or nitric oxide (NO), the molecule is said to be heteronuclear.Can an element be a molecule?
A molecule is the smallest particle of a substance that exists independently. Molecules of most elements are made up of only one of atom of that element. Oxygen, along with nitrogen, hydrogen, and chlorine are made up of two atoms. A compound is a substance formed when two or more elements are chemically joined.Is h2 an element or molecule?
H2, is an elemental gas with an atomic mass of 1.00794. This diatomic molecule is the lightest and most abundant element in the universe. It is also colorless, odorless, and highly flammable. Hydrogen; Hydrogen, H2, is the most abundant element in the universe.Is methane an ionic compound?
Methane and water are composed of molecules; that is, they are molecular compounds. Sodium chloride, on the other hand, contains ions; it is an ionic compound. Ionic compounds contain ions and are held together by the attractive forces among the oppositely charged ions.Why is methane bad?
If methane leaks into the air before being used – from a leaky pipe, for instance – it absorbs the sun's heat, warming the atmosphere. For this reason, it's considered a greenhouse gas, like carbon dioxide.Is hclo4 an ionic compound?
Perchloric AcidWhat are the rules for naming binary molecular compounds?
Naming binary molecular compounds is really quite easy. The first element is given its element name; the second is given its root (hydr, bor, carb, ox, fluor, etc.) followed by ide. For example, HCl is hydrogen chloride, and H2Se is hydrogen selenide.How do you write formulas for ionic compounds?
To write the empirical formula for an ionic compound:- Identify the cation.
- Write the correct formula and charge for the cation.
- Identify the anion.
- Write the correct formula and charge for the anion.
- Combine the cation and anion to produce an electrically neutral compound.
How do you name binary ionic compounds?
Rules for Naming Binary Ionic Compounds- The full name of the cation is listed first.
- The root of the anion name is listed second and is followed by the suffix “ide.”
- If the compound contains a transition metal, a Roman numeral is included after the metal name to indicate the oxidation number of the metal.