Carbon has 4 electrons in it's valence shellwhich makes it a metalloid but commonly it is considered asa non metal..
Beside this, is carbon a metal or metalloid?
Carbon is a nonmetal, and the remainingelements in this group are metals. Group 15 is called thenitrogen group. The metalloids in this group are arsenic andantimony.
Similarly, why carbon is not a metalloid? Carbon doesn't contain the physicalproperties of a metal. Most metals are usually lustrous, opaque,solid, and some are malleable and ductile. Metals are also verygood conductors.
Besides, is carbon considered a metal or a nonmetal?
Carbon (from Latin: carbo "coal") is a chemicalelement with the symbol C and atomic number 6. It isnonmetallic and tetravalent—making four electronsavailable to form covalent chemical bonds. It belongs to group 14of the periodic table.
Is mg a metal nonmetal or metalloid?
The student groups classified magnesium,zinc, iron and tin as metals; sulfur asnonmetal and silicon and carbon as metalloids. Carbonconducts electricity and does not have the characteristicluster.
Related Question Answers
Is oxygen a metal?
No- oxygen is not a metal. Oxygenas you might know it and at room temperature is a gas. Solidoxygen would be available only at extremely lowtemperatures, but, besides that, oxygen does not exhibitmetallic characteristics one might associate with ametal.What are the 8 metalloids?
The metalloids; boron (B), silicon (Si),germanium (Ge), arsenic (As), antimony (Sb), tellurium (Te),polonium (Po) and astatine (At) are the elements found along thestep like line between metals and non-metals of the periodictable.What are the properties of carbon?
Carbon has several allotropes, or different formsin which it can exist. These allotropes include graphite anddiamond, which have very different properties. Despitecarbon's ability to make 4 bonds and its presence in manycompounds, it is highly unreactive under normalconditions.What are the uses of carbon?
Uses of Carbon Carbon (in the form of coal, which is mainlycarbon) is used as a fuel. Graphite is used for pencil tips,high temperature crucibles, dry cells, electrodes and as alubricant. Diamonds are used in jewelry and – because theyare so hard – in industry for cutting, drilling, grinding,and polishing.Is lead a metalloid?
Boron, silicon, germanium, arsenic, antimony, andtellurium are commonly recognised as metalloids. Otherelements are occasionally classified as metalloids. Theseelements include hydrogen, beryllium, nitrogen, phosphorus, sulfur,zinc, gallium, tin, iodine, lead, bismuth, andradon.Why is sodium a metal?
Sodium is an alkali metal, being in group1 of the periodic table, because it has a single electron in itsouter shell, which it readily donates, creating a positivelycharged ion—the Na+ cation. Its only stableisotope is 23Na. The free metal does not occur innature, and must be prepared from compounds.Is carbon a semi metal?
Carbon Family. Of the Group 14 elements, onlycarbon and silicon form bonds as nonmetals (sharingelectrons covalently). Silicon and germanium are semimetals(metalloids), existing in compounds with either +4 or -4 charges.Tin and lead are definitely metals.Is gas a non metal?
Non-metals can be solid, liquid, orgas at room temperature depending upon the element. Sulfur,bromine, and helium are typicalnon-metals.What are the 22 non metals?
Out of 22 non-metals 10non-metals are solid 8 (e.g. carbon, Sulphur andphosphorous), 11 non-metals are gases (e.g. hydrogen,nitrogen, oxygen and chlorine), whereas only onenon-metal (bromine) is a liquidnon-metals.Is Diamond a metal?
Diamond is one of the allotropes of carbon . Itis categorized as Non-metal because of the following points.It is a bad conductor of heat and electricity. Not at all reactiveand does not form oxides.But it has deviations from the propertiesof non-metals.What type of metal is carbon?
Carbon is a non metal, which can be seenby observing its physical properties which are dissimilar tometals. Metals are malleable, ductile like iron andcopper, while carbon is itself found in many forms likediamond(hardest form) , graphite(soft) and the amorphous orpowdery charcoal itself.Is plastic a non metal?
The term metal and non-metals isused for elements. Plastic is not an element but a polymercomposed of different non-metals such as carbon,hydrogen, oxygen, nitrogen etc. Also, not all plastics canbe deformed.Is oxygen a nonmetal?
Nonmetals share many similar propertiesincluding: They are either gas (hydrogen, oxygen, nitrogen)or solid (carbon, sulfur) under standard conditions. They are notgood conductors of electricity or heat. They are very brittle intheir solid form.What is Group 14 on the periodic table called?
The carbon group is a periodic table groupconsisting of carbon (C), silicon (Si), germanium (Ge), tin (Sn),lead (Pb), and flerovium (Fl). In modern IUPAC notation, it iscalled Group 14.Is zinc a metal or nonmetal?
Zinc is classified as a metal element andis a solid at room temperature. It has a melting point of 787degrees Fahrenheit, a boiling point of 1,665 F and an atomic weightof 65.38. The element zinc is assigned the number 30 on theperiodic table of elements, assigned to group number 12 and doesnot have a group name.Is germanium a metalloid?
Germanium is a chemical element with symbol Geand atomic number 32. It is a lustrous, hard, grayish-whitemetalloid in thecarbon group, chemically similar to itsgroup neighbors tin and silicon.Why is Sulphur a non metal?
ABout Sulphur:- Sulfur is characterized asa non-metal because it is consistent with the 3physical properties listed for nonmetals. It is a poor conductor ofheat and electricity because the electrons are not free tomove. Sulfur has a low density because it has less volumefor its electron cloud.Is Gallium a metalloid?
The p-block has its own set of metals located to theleft of the metalloids. These metals are aluminum (Al),gallium (Ga), indium (In), tin (Sn), thallium (Tl), lead(Pb), and bismuth (Bi). These metals are solid, shiny, and goodconductors of electricity and heat, all properties that we normallyassociate with metals. 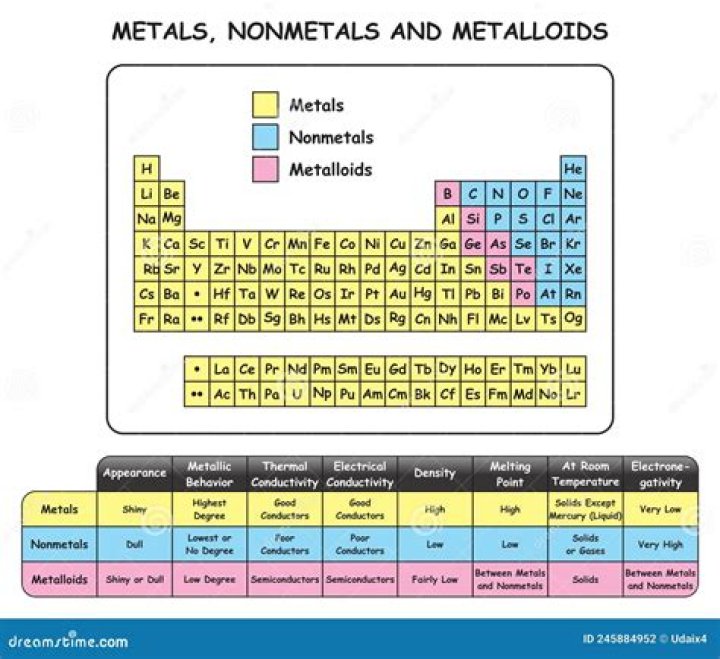
 William Burgess
William Burgess 




