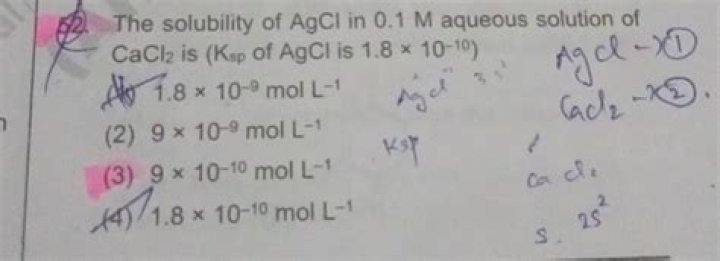
Is AgCl aqueous
 Isabella Bartlett
Isabella Bartlett is agcl aqueous soluble? No. Silver chloride (AgCl) is not soluble and there is very less silver and chloride ions in the aqueous phase.
Is AgCl solid or aqueous solution?
Silver chloride, AgCl, is a white crystalline solid which is well known for its low solubility in water.
What type of solution is AgCl?
Consider a saturated solution of AgCl in water. Because AgCl is a 1:1 salt, the concentrations of the Ag+ and Cl- ions in this solution are equal. Imagine what happens when a few crystals of solid AgNO3 are added to this saturated solution of AgCl in water.
Is AgCl is soluble in water?
It is well established in the pedagogical literature that AgCl is insoluble in water while NaCl and KCl are soluble: applications of this difference are made in elementary studies of both qualitative and quantitative analysis.Is a AgCl soluble or insoluble?
If two solutions are mixed together it is possible that two ions could combine to form an insoluble ionic complex. A solution of silver nitrate is combined with a solution of sodium chloride. The resulting solution contains Na+, Ag+, Cl-, and NO3-, but AgCl is not soluble in water.
What makes AgCl insoluble in water?
Silver chloride (AgCl) do not dissolve in water. The forces holding the solid AgCl lattice together are too strong to be overcome by the forces favoring the formation of the hydrated ions, Ag+(aq) and Cl-(aq). For an ionic compounds to dissolve, the Mandelung energy between ions in the lattice must be overcome.
Is AgCl insoluble salt?
SolubleInsolubleMost common chloridesSilver chlorideSodium, potassium and ammoniumMost common carbonates
Why is AgCl less soluble in seawater?
However, now the amount of silver chloride that will dissolve will be less. This is because there is a common ion in the solution, chloride. … Therefore, in the 0.1 M solution of NaCl only 1.8 x 10-9 moles of AgCl dissolve in one L. This is approximately 10,000 times less than would dissolve in pure water (1.3 x 10-5 M).Is AgCl soluble in water at 25 C?
Answers. The solubility of silver chloride, AgCl, is 1.26 x 10-5 M at 25 °C.
Why is AgCl insoluble in water and soluble in ammonia?AgCl is not soluble in water because the forces holding the solid AgCl lattice together is strong to overcome the forces which favours the formation of hydrated ions. Where as AgCl is soluble in ammonia because it form complexes[Ag(NH)3]2+Cl.
Article first time published onWhat is the solubility product of AgCl?
The solubility product of AgCl is 1.8 × 10^-10 .
Why AgCl is insoluble in nitric acid?
It is nitric acid, in fact. However, AgCl , or silver chloride, is not a base, and so does not form a salt. Surprisingly, AgCl itself is a salt, and a product of a neutralisation reaction: that between hydrochloric acid and silver hydroxide.
Is AgCl or NaCl more soluble?
AgCl is more soluble in aqueous sodium chloride solution than in pure water.
Is AgCl soluble in HCl?
It has been found that the AgCl would readily dissolve in concentrated HCl solution forming AgCln 1−n complexes, as reported by Zelyanskii et al., and the solubility of AgCl increased about 29 times from 0.925 mmol L −1 in 2 M HCl to 26.9 mmol L −1 in 10 M HCl.
Why is AgCl less soluble in NaCl but more soluble in water?
Reason: AgCl dissociates completely and more rapidly than NaCl.
What do you mean by AgCl?
Silver chloride | AgCl – PubChem.
Is AgCl an acid base or salt?
The base of silver is weak, whereas chlorine acid is strong. When the weak base reacts with a strong acid, it forms acidic salt. It means the salt that produces acidic ph, currently (ammonia), becomes a base when dissolved in water. Thus, chlorine from AgCl reacts with NH₃ to produce NH₄-Cl.
Is AgCl a covalent compound?
Hence AgCl is ionic in nature and referred as an ionic compound. … A metal and a non-metal atom forms an ionic bond whereas atoms with less electronegativity will form covalent bonds. The sharing of electrons between the atoms will result in a covalent bond.
Is AgI soluble?
AgI is practically insoluble in water. Only 3 x 10 -7 g will dissolve in 100mL of water at 20 °C.
How do you make an AgCl soluble?
Preparation. Silver chloride is unusual in that, unlike most chloride salts, it has very low solubility. It is easily synthesized by metathesis: combining an aqueous solution of silver nitrate (which is soluble) with a soluble chloride salt, such as sodium chloride or cobalt(II) chloride.
What forces hold AgCl together?
Of the other substances, AgCl is least likely to be a gas because it exists as Ag+ and Cl- ions with very strong ionic bonds holding the ions within the solid.
Why is AgCl less soluble in nacl?
Salt (sodium chloride) nacl is made from positive sodium ions bonded to negative chloride ions. … Agcl is non -polar because of big size of silver ion with chlorine ion and so it’s deform the electrons shells,so this gives its non polar nature so agcl insoluble in water …
Which is more soluble AgCl or AgBr?
AgCl is more soluble in water than AgBr .
What is insoluble in water at 25c?
All silver, lead and mercury(I) salts are insoluble. All chlorides, bromides and iodides are soluble. All carbonates, sulfides, oxides and hydroxides are insoluble. All sulfates are soluble except strontium sulfate and barium sulfate.
What is the concentration of AgCl in an aqueous?
QuestionsAnswerExplanations38 What is the concentration of AgCl in an aqueous solution that contains 1.2 x10-3 gram of AgCl in 800. grams of the solution? (1) 1.2 ppm (3) 7.2 ppm (2) 1.5 ppm (4) 9.6 ppm2use ppm equation on back of RT (0.0012g/800.g) x 1,000,000
Why is AgCl solid?
The forces holding the solid AgCl lattice together are too strong to be overcome by the forces favoring the formation of the hydrated ions, Ag+(aq) and Cl-(aq). Covalently bonded, non polar substances like fats do not dissolve in (pure) water because there is no driving force to form hydrated fat molecules.
Is AgCl more soluble in water or NH3?
AgCl is more soluble in NH3 than in water.
Is AgCl insoluble in NH3?
original precipitateobservationAgIprecipitate is insoluble in ammonia solution of any concentration
How does aqueous ammonia react with AgCl precipitate?
Step 2: When aqueous ammonia is added the silver chloride precipitate dissolves in favor of formation of the silver ammonia complex ion. … The increase in silver ion product concentration pushes the reaction to the left, creating more insoluble silver chloride.
What is the solubility g/l of AgCl?
Example: The solubility of silver chloride is 6.56×10-4 g/L.
What is the molar solubility of AgCl KSP 1.77 10 10?
Sn(OH)2Ksp = 5.45 x 10¯27Ag2CrO4Ksp = 1.12 x 10¯12





