Is actinium rare or common
 Andrew White
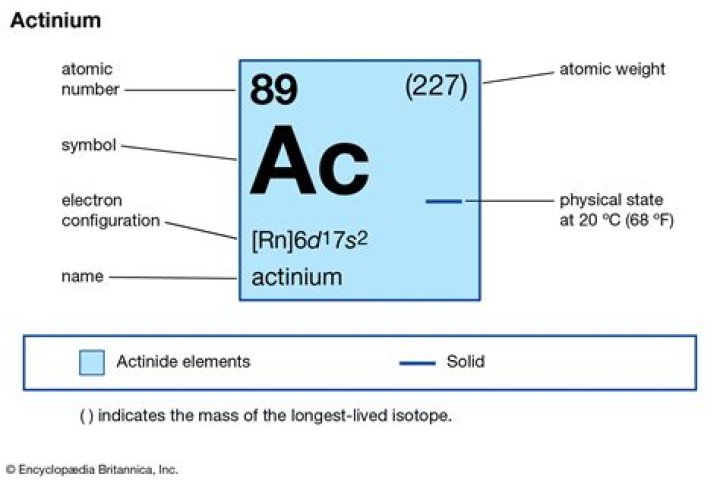
Andrew White Actinium is a rare element that is present in uranium ores in tiny amounts, but it is usually cheaper and easier to create actinium when it is needed by bombarding radium with neutrons in a nuclear reactor. Actinium’s most stable isotope, actinium-227, has a half-life of 21.77 years.
How common is actinium?
Although it is a rare natural element, actinium does occur in uranium ores, where it forms from the radioactive decay of uranium and other radioisotopes, such as radium. Actinium is present at an abundance of 0.0005 parts per trillion by mass in the Earth’s crust.
Which is the rarest element on the Earth?
A team of researchers using the ISOLDE nuclear-physics facility at CERN has measured for the first time the so-called electron affinity of the chemical element astatine, the rarest naturally occurring element on Earth.
Where is actinium most commonly found?
Actinium is found naturally in uranium ores. It rarely occurs as a free element in the earth’s crust. It is more frequently produced in the lab, largely for use as a radiation source. Actinium was first noted as a new element by a French chemist, André-Louis Debierne, in 1899.Why is actinium called actinium?
Word origin: The word actinium comes from the Greek aktis or aktinos, which means beam or ray. Discovery: Actinium has two independent discoverers: Andre Debierne, who found it in 1899, and F. Giesel, who discovered it in 1902.
Where is T on the periodic table?
Name chemical elementSymbolAtomic numberTantalumTa73TechnetiumTc43TelluriumTe52TennessineTs117
Is radium more powerful than uranium?
Radium is about a million times more radioactive than uranium and, under the influence of the heat released, emits an attractive blue colour that Pierre and Marie Curie enjoyed looking at in the evenings. Radium is an extremely rare element that was first discovered in 1898 by Pierre and Marie Curie.
What is 230 on the periodic table?
Thorium-230 | Th – PubChem.What period and group is gold in?
Group111064.18°C, 1947.52°F, 1337.33 KPeriod62836°C, 5137°F, 3109 KBlockd19.3Atomic number79196.967State at 20°CSolid197Au
What is the period of chromium?Group6Melting pointPeriod4Boiling pointBlockdDensity (g cm−3)Atomic number24Relative atomic massState at 20°CSolidKey isotopes
Article first time published onWhat is the 2nd rarest element on earth?
AstatineElectrons per shell2, 8, 18, 32, 18, 7Physical propertiesPhase at STPunknown phaseAtomic properties
Why is gold rare?
Gold is rare throughout the Universe because it’s a relatively hefty atom, consisting of 79 protons and 118 neutrons. That makes it hard to produce, even in the incredible heat and pressure of the ‘chemical forges’ of supernovae, the deaths of giant stars responsible for creating most chemical elements.
What is the rarest thing in the universe?
He designed a rocking horse made of 24-carat gold. Scientists have spotted the “rarest event ever recorded”, in a major breakthrough as part of attempts to solve a dark matter mystery.
What are 3 uses of actinium?
- It is a vital source of alpha rays.
- Ac 225 is used in the field of medicine as an agent for radiation therapy.
- It has immense value as a neutron source as it is one fifty times more radioactive than compared to radium.
- Ac does not find much significant use in any industrial application.
What is MO on the periodic table?
molybdenum (Mo), chemical element, silver-gray refractory metal of Group 6 (VIb) of the periodic table, used to impart superior strength to steel and other alloys at high temperature.
Do radium watches still glow?
Radium dials usually lose their ability to glow in the dark in a period ranging anywhere from a few years to several decades, but all will cease to glow at some point. … The phosphor deterioration means you can’t see a glow anymore, but radium takes thousands of years to completely decay.
What is the most radioactive thing ever?
The radioactivity of radium then must be enormous. This substance is the most radioactive natural element, a million times more so than uranium.
How did radium get banned?
The 1938 Food Drug and Cosmetic Act outlawed deceptive packaging that made Radithor and other radium-branded products marketable.
Is no an element?
nobelium (No), synthetic chemical element of the actinoid series of the periodic table, atomic number 102. The element was named after Swedish chemist Alfred Nobel.
Is sodium man made?
As such sodium is found naturally only in compounds and never as the free element. Even so it is highly abundant, accounting for around 2.6 per cent of the earths crust by weight.
Is there an element that starts with D?
Atomic numberElement symbolElement name110DsDarmstadtium105DbDubnium66DyDysprosium99EsEinsteinium
How did gold get on Earth?
All of the gold found on Earth came from the debris of dead stars. As the Earth formed, heavy elements such as iron and gold sank toward the planet’s core. If no other event had occurred, there would be no gold in the Earth’s crust. But, around 4 billion years ago, Earth was bombarded by asteroid impacts.
Is gold native to Earth?
In its natural form, it is found deep in the layers of the earth where it is transported by water, molten lava and volcanic eruptions, and earthquakes. Geologists have found gold in rocks as old as 4.5 billion years ago.
What element is closest to gold?
OsmiumIridiumPlatinumGoldMercury (element)
Is thorium named after Thor?
Discovery date1829Origin of the nameThorium is named after Thor, the Scandinavian god of war.Allotropes
Is thorium man made?
Thorium (chemical symbol Th) is a naturally occurring radioactive metal found at trace levels in soil, rocks, water, plants and animals. … There are natural and man-made forms of thorium, all of which are radioactive. In general, naturally occurring thorium exists as Th-232, Th-230 or Th-228.
How is thorium 232 used?
Thorium is used as an alloying agent to improve magnesium’s strength at high temperatures. … When bombarded with neutrons, thorium-232 becomes thorium-233, which eventually decays into uranium-233 through a series of beta decays. Uranium-233 is a fissionable material and can be used as a nuclear fuel.
What is chromium family name?
Group 6, numbered by IUPAC style, is a group of elements in the periodic table. Its members are chromium (Cr), molybdenum (Mo), tungsten (W), and seaborgium (Sg). These are all transition metals and chromium, molybdenum and tungsten are refractory metals.
What Colour is chromium?
Chromium compounds are all vividly colored and are used as pigments — bright green, yellow, red and orange. Rubies are red because of chromium, and glass treated with chromium has an emerald green color, according to the Royal Society of Chemistry (RSC).
What makes chromium unique?
Chromium is a lustrous, brittle, hard metal. Its colour is silver-gray and it can be highly polished. It does not tarnish in air, when heated it borns and forms the green chromic oxide. Chromium is unstable in oxygen, it immediately produces a thin oxide layer that is impermeable to oxygen and protects the metal below.
What elements are not found on Earth?
There are two elements with no stable isotopes and fairly short half-lives and hence are not found on Earth, since any that may have been here at some time have long since decayed to other (more) stable elements. Those elements are Technetium (atomic number 43) and Promethium (atomic number 61).