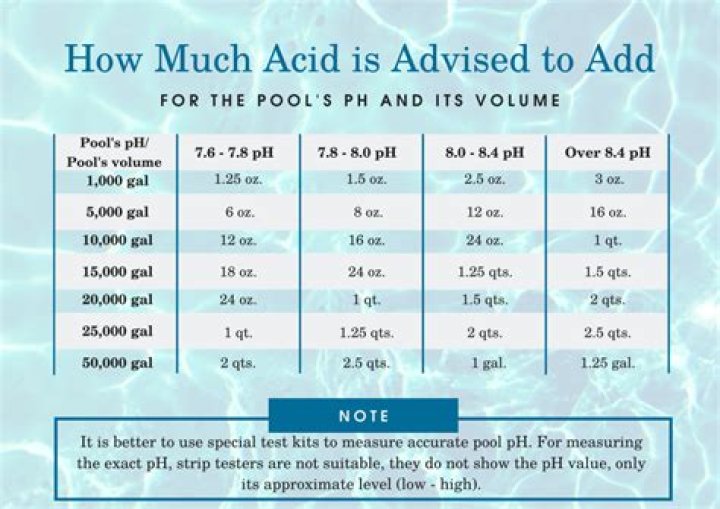
How much acid do I add to lower pH?
 Emma Terry
Emma Terry .
Moreover, how much acid is needed to change the pH of water?
Diluting acids and bases To make the pH change by 1, a tenfold dilution is required (eg adding 9 cm 3 of water to 1 cm 3 acid). The acid is becoming less acidic.
Secondly, how do acids affect pH? When added to water, an acid releases hydrogen ions. The CO2 breaks down to carbonic acid that then dissociates to release hydrogen ions into the water, decreasing the pH. To answer your question, acid added to water increases the hydrogen ion concentration (H+), decreases the pH, and increases the acidity.
Also to know is, does adding acid raise or lower pH?
To raise or lower pH, a pool custodian simply adds acids or alkalis into the water. For example, adding sodium carbonate (soda ash) or sodium bicarbonate (baking soda) will generally raise the pH, and adding muriatic acid or sodium bisulfate will lower the pH.
How do I lower my pH level?
Pure or distilled water has a pH level of 7, which means it is neutral. If you want to increase the pH of water, you must add an alkaline substance, such as baking powder, to it. If you want to decrease the pH of water, you add an acidic substance, such as lemon juice, to it.
Related Question AnswersHow do you change the pH of water?
If you've measured your tap water and found that it's basic (or too acidic), you'll need to adjust the water's pH level before watering your plants. Lime or wood ash can be added to a 1 gallon (3.8 L) container of water to raise the pH. To lower the pH, add phosphoric acid or a tablet designed to lower water pH.How do you adjust the pH level in water?
Soda ash/sodium hydroxide injection This treatment method is used if water is acidic (low pH). Soda ash (sodium carbonate) and sodium hydroxide raise the pH of water to near neutral when injected into a water system. Unlike neutralizing filters, they do not cause hardness problems in treated water.Does adding chlorine increase pH?
Using liquid chlorine raises the pH of the water. Liquid chlorine does not raise pH. When added to water, liquid chlorine (which has a pH of 13) makes HOCl (hypochlorous acid – the killing form of chlorine) and NaOH (sodium hydroxide), which raises pH. So the net effect on pH is zero (or almost zero).Will vinegar lower pH in pool?
Lower the pH of the water by measuring out four cups of vinegar and pouring it directly into the waters. You can use either white household vinegar or apple cider vinegar.What is the pH of water?
pH and Water The pH of pure water is 7. In general, water with a pH lower than 7 is considered acidic, and with a pH greater than 7 is considered basic. The normal range for pH in surface water systems is 6.5 to 8.5, and the pH range for groundwater systems is between 6 to 8.5.Is NaCl an acid or base?
NaCl is formed by the reaction of HCl and NaOH. Both are strong acids and bases. When a strong acid and a strong base react together the resultant is salt and water. Therefore NaCl is a salt.How do you maintain a pH meter?
For most pH sensors, it's critical that the sensor be stored wet in the appropriate solution. To clean most pH sensors, rinse in distilled or deionized water. Shake off any excess water and return the sensor to its storage solution. The majority of pH sensors have a lifespan of approximately 1-2 years.What is the pH of a very strong acid?
Strong Acids Generally, a strong acid has a pH of about zero to 3. The stronger the acid, the better it dissociates in an aqueous solution, releasing more cationic hydrogen (H+) ions.Why is the pH in my pool always high?
An increase in the pH level can be caused by several things. A first cause is the addition of chlorine stabiliser or HTH Granular to the pool water. A higher pH level can also be caused by a sudden rise in temperature of the water. However, much more important than the causes are the effects of such a high pH level.Does shock raise pH?
Chlorine based pool shock (Calcium Hypochlorite) has a high pH, and will naturally raise the pH level of your swimming pool water, in addition to changing your chlorine level. Chlorine free shock has a neutral pH, and will not affect any of your pool chemical levels.What happens if pool pH is too high?
If the pH gets higher than 7.8, the water is becoming too alkaline. When water is too alkaline, it reduces the effectiveness of the chlorine — the pool chemical that kills pathogens. Water with a pH that's too high also can cause skin rashes, cloudy water and scaling on pool equipment.What is the pH of Sulphuric acid?
Originally Answered: What is the pH value of sulphuric acid? The Ph is around 2. It could be 1 if it is even more concentrated. Only super concentrated H2SO4 would have a Ph value of 7, because it doesn't have water & therefore cannot really be called an acid.Is high pH water good for you?
Because alkaline water has a higher pH level than does plain tap water, proponents say that it can neutralize acid in your bloodstream. Some say that alkaline water can help prevent disease, such as cancer and heart disease.How long after adding muriatic acid can you swim?
Muriatic acid can create a hot spot of acid in the water that could potentially burn or irritate your skin. It is best to wait 30 minutes after adding it to your pool. We recommend waiting at least 15 minutes to swim after adding algaecide to your swimming pool.What is the fastest way to lower pH in soil?
If your soil is alkaline, you can lower your soil's pH or make it more acidic by using several products. These include sphagnum peat, elemental sulfur, aluminum sulfate, iron sulfate, acidifying nitrogen, and organic mulches.What is the pH of hydrochloric acid?
It consists mainly of hydrochloric acid and acidifies the stomach content to a pH of 1 to 2.What is a good pH level?
The human body is built to naturally maintain a healthy balance of acidity and alkalinity. The lungs and kidneys play a key role in this process. A normal blood pH level is 7.40 on a scale of 0 to 14, where 0 is the most acidic and 14 is the most basic.What has a pH of 8?
What does it mean for a solution to be acidic or basic (alkaline)?| pH Value | H+ Concentration Relative to Pure Water | Example |
|---|---|---|
| 5 | 100 | black coffee, bananas |
| 6 | 10 | urine, milk |
| 7 | 1 | pure water |
| 8 | 0.1 | sea water, eggs |