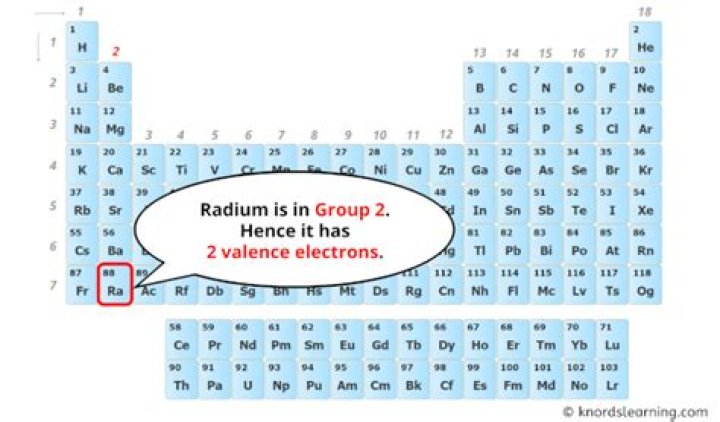
How many valence electrons does radium have
 John Peck
John Peck Radium, Ra, has two valence electrons.
How many electron shells does radium have?
Classification:Radium is an alkali earth metalProtons:88Neutrons in most abundant isotope:138Electron shells:2,8,18,32,18,8,2Electron configuration:[Rn] 7s2
How many core electrons are in radium?
Radium atoms have 88 electrons and the shell structure is 2.8. 18.32. 18.8.
Does radon have 4 valence electrons?
Radon has 8 valence electrons. Radon is a radioactive element, which means it is unstable and undergoes radioactive decay. … It only has 2 valence electrons.What's the atomic number for radium?
With atomic number 88, it has four natural isotopes of atomic weight 228, 226, 224 and 223 – though there are a remarkable 21 more artificial isotopes.
What is BA in the periodic table?
barium (Ba), chemical element, one of the alkaline-earth metals of Group 2 (IIa) of the periodic table.
Where do you find radium?
Where is radium found? Radium was first found in Bohemia in the rich pitchblence ore. Some can also be found in the Carnotite sands of Colorado, although richer supplies exist in regions of Zaire, Africa and the Great Bear Lake region of Canada.
How many electrons have an N 4 in radon?
Radon Atomic and Orbital Properties Radon atoms have 86 electrons and the electronic shell structure is [2, 8, 18, 32, 18, 8] with Atomic Term Symbol (Quantum Numbers) 1S0.What type of metal is radium?
radium (Ra), radioactive chemical element, the heaviest of the alkaline-earth metals of Group 2 (IIa) of the periodic table. Radium is a silvery white metal that does not occur free in nature.
How do you determine valence electrons?Valence electrons can be found by determining the electronic configurations of elements. Thereafter the number of electrons in the outermost shell gives the total number of valence electrons in that element.
Article first time published onHow many neutrons does radium have?
A radium nucleus is a massive nucleus of 226 nucleons, including 88 protons and 138 neutrons.
What does radium look like?
Radium is silvery, lustrous, soft, intensely radioactive. It readily oxidizes on exposure to air, turning from almost pure white to black. Radium is luminescent, corrodes in water to form radium hydroxide. Although is the heaviest member of the alkaline-earth group it is the most volatile.
How many valence electrons do have?
Iodine has seven valence electrons.
How is radium formed?
Radium is a radionuclide formed by the decay of uranium and thorium in the environment. The most common isotopes. For example, uranium has thirty-seven different isotopes, including uranium-235 and uranium-238. of radium are Ra-226 and Ra-228.
Is radium banned?
Radium was eventually banned after scores of dial painters died from cancer and various ghastly ailments. But many of the so-called radium watches are still around today, considered antiques and even prized as collectibles.
How did Marie Curie extract radium?
Marie extracted pure radium salts from pitchblende, a highly radioactive ore obtained from mines in Bohemia. The extraction required tons of the substance, which she dissolved in cauldrons of acid before obtaining barium sulphate and other alkalines, which she then purified and converted into chlorides.
What does RN mean in electron configuration?
ElementTerm SymbolRadium[Rn] 7s21S0Radon[Xe] 4f14 5d10 6s2 6p61S0Rhenium[Xe] 4f14 5d5 6s26S5/2Rhodium[Kr] 4d8 5s14F9/2
What is the shorthand notation for radium?
Radium is a chemical element with symbol Ra and atomic number 88.
How much is radium worth?
Because of its rarity and high demand, radium had become the most expensive material in the world: in equal mass, it was worth more than diamond. One gram of radium costed $ 100,000 at the time (a bit less than 2 million euros in 2018).
What is Br and Ba in the periodic table?
The design highlights two elements on the Periodic table, “Br” and “Ba.” The first is the symbol for Bromine, a chemical element which is useful for fire retardants. The second stands for Barium, a chemical element used frequently in fireworks.
Which element is a metal BA or at?
BariumPhase at STPsolidMelting point1000 K (727 °C, 1341 °F)Boiling point2118 K (1845 °C, 3353 °F)Density (near r.t. )3.51 g/cm3
What is the group number of BA in periodic table?
Group2727°C, 1341°F, 1000 KPeriod61845°C, 3353°F, 2118 KBlocks3.62Atomic number56137.327State at 20°CSolid138Ba
What things have radium in them?
- In Chocolate. Mental Floss. …
- In Water. Wikimedia/Jacopo Werther. …
- In Toys And Nightlights. Mental Floss. …
- In Toothpaste. Mental Floss. …
- In Cosmetics. Mental Floss. …
- In Heating Pads And Suppositories. Orau.org. …
- In The Treatment Of Impotence. …
- In Health Spas.
Is radium and radon the same?
Not exactly. For starters, radon is a gas and radium is a solid. Radium is produced by the natural decay of uranium, a radioactive element found in nature in low quantities.
Is radium the same as radiation?
As nouns the difference between radiation and radium is that radiation is the shooting forth of anything from a point or surface, like the diverging rays of light; as, the radiation of heat while radium is a radioactive metallic chemical element (symbol ra) with an atomic number of 88.
Is radium and uranium the same?
All three forms of uranium have the same chemical and physical properties, but have different radioactive properties. … As uranium decays naturally over time, it releases radiation and forms new elements like radium, lead, and radon gas. Radium occurs naturally from the radioactive decay of uranium over time.
How many electrons can n 2 have?
(a) When n = 2, there are four orbitals (a single 2s orbital, and three orbitals labeled 2p). These four orbitals can contain eight electrons. Again, each orbital holds two electrons, so 50 electrons can fit in this shell.
What is group valence?
Valence refers to the ability of an atom or a group of chemical bonded atoms to form chemical form with other atoms or groups of atoms. The valency of an element is determined by the number of outer shell (valence) electron.
What is an example of a valence electron?
Valence electrons are the electrons in the outermost shell, or energy level, of an atom. For example, oxygen has six valence electrons, two in the 2s subshell and four in the 2p subshell.
How do you find the valence of an element?
valence = number of electrons in valence shell of free atom – number of non-bonding electrons on atom in molecule, valence = number of bonds + formal charge.
What element contains 84 protons?
Polonium, (element 84), was discovered in 1898 and named after Poland, the homeland of Marie Curie (Ne Sklodowska) who found it with her husband Pierre Curie.