How many molecules of ammonia are in 2nh3
 Christopher Lucas
Christopher Lucas 2NH3. Question 17 options: 6 nitrogen and 6 hydrogen. 2 nitrogen and 3 hydrogen.
How many atoms and molecules are present in 2NH3?
2NH3. Question 17 options: 6 nitrogen and 6 hydrogen. 2 nitrogen and 3 hydrogen.
What is 2NH3?
You would use a designation like “2NH3” only if you were writing a chemical equation, such as: N2 + 3H2 → 2NH3. The coefficients indicate that one molecule of nitrogen (N2) reacts with three molecules of hydrogen (H2) to make two molecules of ammonia (NH3).
How many molecules of ammonia are in NH3?
This means that 17g of ammonia is one mole of ammonia. The number of molecules in a single mole of any compound is given by Avoadro’s constant, which is 6.023×10^23. There would be 6.023×10^23 molecules of ammonia in 17 grams of the substance.What compound is 2NH3?
NamesChemical formulaNH3Molar mass17.031 g/molAppearanceColourless gasOdorstrong pungent odour
What is the molar mass of 2nh3?
17.03052 g/mol .
How many ammonia molecules are produced?
Answer: 1 molecule of nitrogen (N2) are used to produce two molecules of ammonia (NH3).
How many molecules of ammonia are there in 5 moles of NH3?
Avogadro said that 1 mole of any compound will contain 6.023 × 10²³ particles. That implies, 5 moles of NH3 will contain 5 × 6.023 × 10²³ NH3 molecules.What is the molecular structure of NH3?
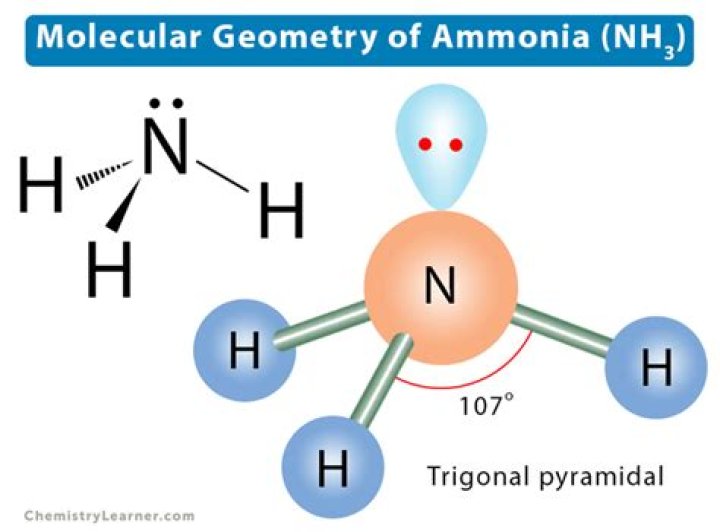
The ammonia molecule has a trigonal pyramidal shape with the three hydrogen atoms and an unshared pair of electrons attached to the nitrogen atom.
What is N2 3H2 → 2NH3?N2+3H2=2NH3. Nitrogen + Hydrogen = Ammonia. This is an example of a combination reaction because in this combination reaction two smaller molecules combined together to form a bigger compound molecule. A combination reaction is in which two reactants combine to form one product.
Article first time published onHow many molecules are in 34g of ammonia?
therefore 34 g of NH₃ contains 12.044*10²³ molecules .
Is NH3 ionic or covalent?
NH3 has a covalent single bond among its nitrogen and hydrogen atoms. A covalent bond means the N and H atoms share valence electrons while creating…
How do you calculate molecules produced?
Determine the mass of the substance, and its molar mass. Divide the given mass by its molar mass to get moles, then multiply times 6.022×1023molecules1mol .
How many NH3 molecules are produced from the three N2?
Complete answer: N2 undergoes a reduction to form two molecules of NH3. During this reaction, 16 molecules of ATP are hydrolysed to form two molecules of ammonia.
How many molecules does ammonia have on the reactant side?
We see that 1 molecule of nitrogen reacts with 3 molecules of nitrogen to form 2 molecules of ammonia. This is the smallest possible relative amounts of the reactants and products.
What is molecular mass of co2?
The molecular mass of carbon dioxide is 44.01amu. The molar mass of any compound is the mass in grams of one mole of that compound. One mole of carbon dioxide molecules has a mass of 44.01g, while one mole of sodium sulfide formula units has a mass of 78.04g. The molar masses are 44.01g/mol and 78.04g/mol respectively.
Is ammonia an ion?
SynonymsSourcesammonium ionPDBeChemAmmonium(1+)ChemIDplusNH4+KEGG COMPOUNDNH4+IUPAC
What is the charge of ammonia?
Ammonia has zero charge because it is a neutral molecule that consists of a nitrogen atom bonded to three hydrogen atoms by covalent bonds. There is an unshared pair of electrons over the N-atom in ammonia.
How many molecules are in 10 moles of ammonia?
10 mol NH3 contains 6×1024molecules NH3 .
How many molecules of NH3 are in 8.8 moles?
Step 2: Find the Number of Moles The example is 60.50 grams of CaCl2. Change this into moles using the molar mass you found in Step 1. Chemists use ratios for this calculation.
How can you represent 2 molecules of ammonia?
- This shows that ammonia exists as diatoms. …
- To achieve this, nitrogen shares three of its electrons with three hydrogen atoms. …
- This separate pair of electrons of one is shared with the lone pair of another molecule to form a stable compound.
What is the coefficient of N2 3H2 → 2NH3?
Nitrogen has no coefficient so it’s a 1, and Hydrogen has a 3. The ratio of Hydrogen to Nitrogen is therefore 3 to 1.
What is the enthalpy of N2 3H2 2NH3?
The enthalpy change for the reaction N2+3H2 gives 2NH3 is -92.32 kJ at 298 K.
How many moles of NH3 molecules are there in 6.8 g NH3?
Thus correct answer is 0.5 moles.
How many molecules are in 36 grams of ammonium?
Hey! The formula mass of ammonia is 14 + 1 3 = 17. The number of moles in 36 g ammonia is 36/ 17 = 2.11mol.
How many molecules are there in 1.8 gram of water?
1.8 gram of water will contain 0.1×6.023×1023 number of molecules.
What type of compound is ammonia NH3?
Ammonia, NH3, is a chemical compound composed of one nitrogen atom and three hydrogen atoms. Ammonia is a colorless gas that is lighter than air, and can be easily liquefied. Ammonia is a compound normally encountered as a gas and has a strong odor.
What type of bond is NH3?
Ammonia (NH3) has polar covalent bond.
Is ammonium an ionic or molecular compound?
NH3 or commonly known as Ammonia is held together with covalent bonds, not ionic. This means that they’re sharing electron pairs. The molecule is polarized with a slight positive charge on the H3 side and conversely a slight negative charge on the Nitrogen side. NH3, also known as ammonia is a covalent compound.
How do you find the number of ions?
Number of mol is calculated by ratio of given mass to the molar mass. After calculating the number of moles, the number of ions will be equal to the product of the number of moles and Avogadro’s number.
How many molecules are in 1.5 moles?
Hence, number of molecules in 1.5 moles of ammonia is 9.033 × 1023.