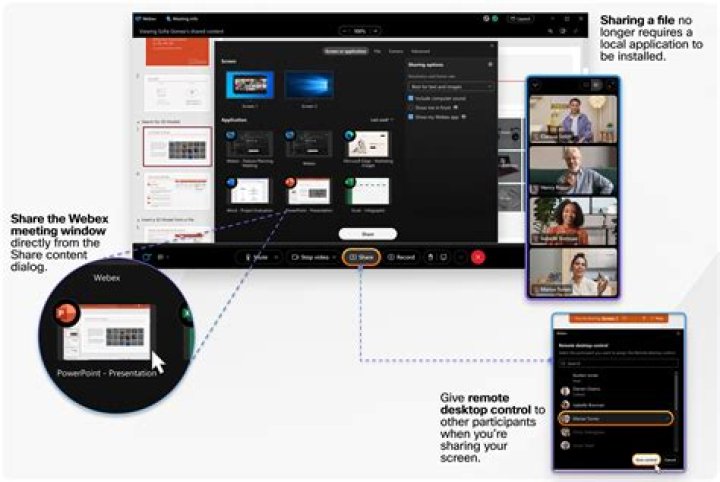
How many molecules are in 5.2 moles of h2o?
 William Burgess
William Burgess .
Accordingly, how many molecules are in 2.5 moles of h20?
Explanation: You can calculate the number of molecules from moles by using Avogadro's constant, 6.02×1023 molecules/mol . For every 1 mol, there are 6.02×1023 molecules.
Additionally, how many molecules are in 2 moles of water? 1 mole = 6.022×10^23 atoms. 1 water molecule = 2 Hydrogen atoms + 1 oxygen atom. So, 1 mole H2O = 1.2044×10^24 hydrogen atoms. Therefore 2 mole H2O will have 2.4088×10^24 hydrogen atoms.
Furthermore, how many molecules are in 1.5 moles of h2o?
( 1/4 of a mole) x (6.02 x 1023 atoms/mole) = approximately 1.5 x 1023 atoms. If you have a compound like H2O, then: one mole of water contains 6.02 x 1023 MOLECULES of water. But each molecule of water contains 2 H and 1 O atom = 3 atoms, so there are approximately 1.8 x 1024 atoms in a mole of water.
How many molecules is 5 moles?
This number, which can only be found by practical measurement, is called Avagadro's constant/number, often denoted by N(A). It is about 6 x 10^23. So in 5 moles of oxygen gas, which has a mass of 5x16=80 grams, there are 5x6x10^23 = 30x10^23 molecules.
Related Question AnswersHow many grams are in 2.5 moles of h2o?
1 mole of hydrogen atoms got 1 gram, so 2 moles of them got 2 grams. 1 mole of oxygen atoms got 16 grams. Therefore, 1 mole of H2O got 18 grams.How many atoms are in 2.5 moles?
There are 45.165 X 1023 atoms in 2.5 moles of SO2. We reach this conclusion by using Avogadro's number which is the number of fundamental units in aHow many moles are in 5.80 moles?
Terms in this set (24) fluorine is a diatomic molecule so multiply by 2. How many atoms are in 5.80 moles of Helium? 5.80 mol He 6.02210^23 atoms He/ 1 mol He. This cancels out moles, leaving 3.49*10^24 atoms He.What is the mass of 2.5 moles of water?
A mole is a convenient counting unit whenever one is dealing with numbers of atoms or molecules. It is equal to Avogadro's number (NA), namely 6.022 x1023. If we have one mole of water, then we know that it will have a mass of 2 grams (for 2 moles of H atoms) + 16 grams (for one mole O atom) = 18 grams.How many molecules are in a mole?
Avogadro's number is a very important relationship to remember: 1 mole = 6.022×1023 6.022 × 10 23 atoms, molecules, protons, etc. To convert from moles to atoms, multiply the molar amount by Avogadro's number. To convert from atoms to moles, divide the atom amount by Avogadro's number (or multiply by its reciprocal).How many molecules of sugar c6h12o6 are in a mole?
Answer and Explanation: 1 mole of glucose gives 12 moles of Hydrogen (C6H12O6). So, x/180 moles of glucose gives x/15 moles of hydrogen. Now, according to Avogadro, 1 mole of atom or molecule has 6.023 * 10^23 numbers of that atoms or molecules.What is the point of Avogadro's number?
Avogadro's number is a proportion that relates molar mass on an atomic scale to physical mass on a human scale. Avogadro's number is defined as the number of elementary particles (molecules, atoms, compounds, etc.) per mole of a substance. It is equal to 6.022×1023 mol-1 and is expressed as the symbol NA.How many atoms are in a compound?
Explanation: There are 2 Oxygen atoms (the subscript), and 1 Nitrogen atom. So yes, in total there are 3 atoms.How many grams are in 5 moles of h2o?
From this you can see that 5 moles of hydrogen gas would react with 2.5 of the available 3 moles of oxygen gas, to form 5 moles of water. The molar mass of water is 18 g/mol, so you would end up with 5 x 18 = 90 g of water.How many moles are in a gram?
The answer is 0.0087094358027487. We assume you are converting between moles In and gram. You can view more details on each measurement unit: molecular weight of In or grams The SI base unit for amount of substance is the mole. 1 mole is equal to 1 moles In, or 114.818 grams.How many moles are in 16 g of o2?
Now, each mole of a substance contains an Avogadro's number (or, ) of particles. In other words, 1 mole of oxygen would contain molecules. Here, we are given with 16 g of oxygen. The number of molecules in 16 gm of oxygen are 0.5 (= 16/32 moles).How do I calculate moles?
- Start with the number of grams of each element, given in the problem.
- Convert the mass of each element to moles using the molar mass from the periodic table.
- Divide each mole value by the smallest number of moles calculated.
- Round to the nearest whole number. This is the mole ratio of the elements and is.