Thermochemistry is the part of thermodynamics that studies the relationship between heat and chemical reactions. Thermochemistry is a very important field of study because it helps to determine if a particular reaction will occur and if it will release or absorb energy as it occurs..
Regarding this, what is the relationship between thermochemistry and thermodynamics?
Thermochemistry is the study and measurement of heat energy associated with chemical reactions. Thermodynamics is the branch of physical science that deals with the relations between heat and other forms of energy. Thermochemistry describes the relationship between heat energy and chemical reactions.
Secondly, how does thermochemistry relate to real life? Uses and Examples From the simple things such as putting ice into your glass of water to the common such as burning fuel for a car. When one exercises, the body naturally cools down due to sweating. That is because our bodies supply the heat necessary to evaporate the water.
Beside above, is thermochemistry the same as thermodynamics?
Thermodynamics is the study of the relationship between heat, work, and other forms of energy. Thermochemistry is a branch of thermodynamics which is the study of heat given off or absorbed in a chemical reaction.
What does thermodynamics mean in chemistry?
From Wikipedia, the free encyclopedia. Chemical thermodynamics is the study of the interrelation of heat and work with chemical reactions or with physical changes of state within the confines of the laws of thermodynamics.
Related Question Answers
How do you define enthalpy?
Enthalpy is a thermodynamic property of a system. It is the sum of the internal energy added to the product of the pressure and volume of the system. It reflects the capacity to do non-mechanical work and the capacity to release heat. Enthalpy is denoted as H; specific enthalpy denoted as h.What is the importance of thermochemistry?
Thermochemistry is the part of thermodynamics that studies the relationship between heat and chemical reactions. Thermochemistry is a very important field of study because it helps to determine if a particular reaction will occur and if it will release or absorb energy as it occurs.Why is it important to study thermodynamics?
It's of practical importance because it governs the behavior of chemical reactions and processes that convert energy in the form of heat into other forms of energy, like mechanical work or electrical potentials.What are the three laws of thermodynamics?
The first law, also known as Law of Conservation of Energy, states that energy cannot be created or destroyed in an isolated system. The third law of thermodynamics states that the entropy of a system approaches a constant value as the temperature approaches absolute zero.How do you calculate Q in thermodynamics?
Since the system has constant volume (ΔV=0) the term -PΔV=0 and work is equal to zero. Thus, in the equation ΔU=
q+w w=0 and ΔU=
q. The internal energy is equal to the heat of the system.
Introduction.
| Process | Sign of heat (q) | Sign of Work (w) |
| Heat released from the system- exothermic (absorbed by surroundings) | - | N/A |
What is an example of the first law of thermodynamics?
The first law of thermodynamicsShown are two examples of energy being transferred from one system to another and transformed from one form to another. Humans can convert the chemical energy in food, like this ice cream cone, into kinetic energy by riding a bicycle.What is the difference between enthalpy and internal energy?
Enthalpy: Enthalpy is the heat energy that is being absorbed or evolved during the progression of a chemical reaction. Internal Energy: Internal energy of a system is the sum of potential energy and kinetic energy of that system.What is thermochemistry Why is it important quizlet?
thermochemistry: the relationship between chemistry and energy. It's important because energy and its uses are important to society. It's important to understand how much energy is required or released in a process. law of conservation of mass: energy cannot be created or destroyed.What are the applications of thermochemistry?
Thermochemistry is useful in predicting reactant and product quantities throughout the course of a given reaction. In combination with entropy determinations, it is also used to predict whether a reaction is spontaneous or non-spontaneous, favorable or unfavorable.What is concept of entropy?
Entropy, the measure of a system's thermal energy per unit temperature that is unavailable for doing useful work. Because work is obtained from ordered molecular motion, the amount of entropy is also a measure of the molecular disorder, or randomness, of a system.What are the principles of thermochemistry?
Two fundamental principles of thermochemistry are: (i) the energy change associated with any process is equal and opposite to the reverse process (due to Laplace), and (ii) the energy change for a series of stepwise processes or reactions is the same as that of the entire process (Hess' law).What is calorimetry used for?
A calorimeter is an object used for calorimetry, or the process of measuring the heat of chemical reactions or physical changes as well as heat capacity. Differential scanning calorimeters, isothermal micro calorimeters, titration calorimeters and accelerated rate calorimeters are among the most common types.What is the difference between heat and temperature?
Often we think that heat and temperature are the same thing. However, this is not the case. Heat and temperature are related to each other, but are different concepts. Heat is the total energy of molecular motion in a substance while temperature is a measure of the average energy of molecular motion in a substance.What is meant by heat capacity?
Heat capacity or thermal capacity is a physical property of matter, defined as the amount of heat to be supplied to a given mass of a material to produce a unit change in its temperature. The SI unit of heat capacity is joule per kelvin (J/K). Heat capacity is an extensive property.What is Hess's Law equation?
The enthalpy change for the overall process is the sum of the enthalpy change of the steps in the process. This is known as Hess's Law and is given in the following equation. ΔHrxn=ΔH1+ΔH2+ΔH3+⋯What is heat in thermochemistry?
Heat. Heat is the transfer of energy that results from the difference in temperature between a system and its surroundings. The field of study is called thermochemistry, and it is used to assess the efficacy of fuels, the energy flow in chemical plants, and the strengths of chemical bonds.How is thermodynamics used today?
Sweating in a Crowded Room The human body obeys the laws of thermodynamics. Heat from your body is transferred to the sweat. As the sweat absorbs more and more heat, it evaporates from your body, becoming more disordered and transferring heat to the air, which heats up the air temperature of the room.How does heat transfer affect our daily lives?
FEW EXAMPLES IN DAY TO DAY LIFE ARE - Heat from the iron is conducted to the shirt, making it easy to iron out all those unsightly wrinkles and make the shirt look sharp. The engine of a car is turned on, and the hood becomes warm due to the conduction of heat from the engine to the hood of the car.What does the second law of thermodynamics mean?
The First Law of Thermodynamics states that energy cannot be created or destroyed; the total quantity of energy in the universe stays the same. The Second Law of Thermodynamics is about the quality of energy. It states that as energy is transferred or transformed, more and more of it is wasted. 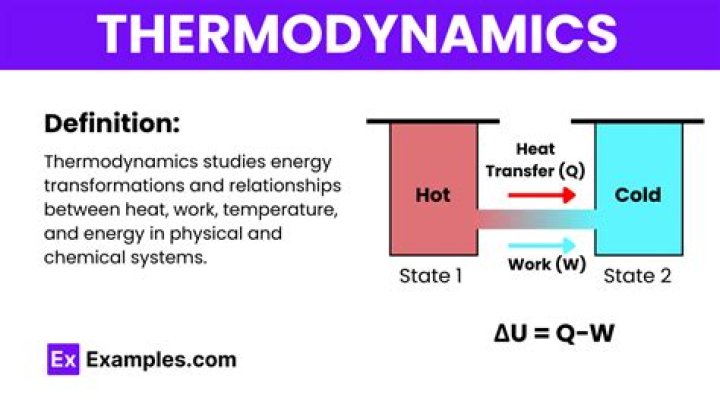
 William Burgess
William Burgess