Answer and Explanation: The ideal gas law is usually written as PV = nRT. In this formula: P stands for pressure in atmospheres (atm). V stands for volume in liters (L)..
Keeping this in view, how is the ideal gas law derived?
The ideal gas law is derived from empirical relationships among the pressure, the volume, the temperature, and the number of moles of a gas; it can be used to calculate any of the four properties if the other three are known.
Also, can an ideal gas be condensed? Since the particles of an ideal gas have no volume, a gas should be able to be condensed to a volume of zero. Reality check: Real gas particles occupy space. As kinetic energy decreases as a gas is cooled, the particles will eventually move slowly enough that there attractive forces cause them to condense.
Also question is, what is an ideal gas example?
An ideal gas is a theoretical gas composed of many randomly moving point particles whose only interactions are perfectly elastic collisions. Many gases such as nitrogen, oxygen, hydrogen, noble gases, and some heavier gases like carbon dioxide can be treated like ideal gases within reasonable tolerances.
What does the ideal gas law allow a scientist?
The ideal gas law allows the scientists to calculate the number of moles of the gas whereas other gas laws deal with the temperature, pressure and volume of the gas only. Further Explanation: The Boyle's Law states that at constant temperature, the pressure of a gas is inversely proportional to the volume of the gas.
Related Question Answers
Why is the ideal gas law important?
Boyle's Law states that, when temperature is constant, the pressure and volume of a fixed sample of a gas are inversely proportional (P1 x V1 = P2 x V2). So the significance is that the ideal gas law calculates the behavior of any gas under common conditions of temperature and pressure.What is the formula for the ideal gas law?
nH2 = PH2V / RT ; nH2 = (0.9503 atm)(0.456 L) / (0.0821 L-atm / mole-K)(295 K) = 0.0179 mole H2. The ideal gas equation (PV=nRT) provides a valuable model of the relations between volume, pressure, temperature and number of particles in a gas. As an ideal model it serves as a reference for the behavior of real gases.Who Discovered ideal gas law?
Benoît Paul Émile Clapeyron
What is ideal gas law definition?
ideal gas law. noun. A physical law describing the relationship of the measurable properties of an ideal gas, where P (pressure) × V (volume) = n (number of moles) × R (the gas constant) × T (temperature in Kelvin). It is derived from a combination of the gas laws of Boyle, Charles, and Avogadro.Who made the ideal gas law?
Benoît Paul Émile Clapeyron
How is Boyle's law derived from ideal gas law?
AVOGADRO'S LAW states the volume of a gas is directly proportional to the number of moles. BOYLE'S LAW states that the volume of a gas is inversely proportional to its pressure. CHARLES'S LAW states that the volume of a gas is directly proportional to its Kelvin temperature.What are the three laws of gas?
The gas laws consist of three primary laws: Charles' Law, Boyle's Law and Avogadro's Law (all of which will later combine into the General Gas Equation and Ideal Gas Law).What is real gas equation?
Originally, the ideal gas law looks like this: PV = nRT. P is the pressure in atmospheres, V is the volume of the container in liters, n is the number of moles of gas, R is the ideal gas constant (0.0821 L-atm/mol-K), and T is the temperature in Kelvin.What are the characteristics of an ideal gas?
The properties of an ideal gas are: An ideal gas consists of a large number of identical molecules. The volume occupied by the molecules themselves is negligible compared to the volume occupied by the gas. The molecules obey Newton's laws of motion, and they move in random motion.Is ch4 an ideal gas?
Methane (CH4) behaves as an ideal gas under standard temperature and pressure conditions. What volume is occupied by 1 kg of methane at a temperature of 35 °C and a pressure of 0.9 atm?What is an ideal or perfect gas?
Perfect gas is one in which intermolecular forces are not considered. Most of the gases behave as perfect gases at low pressures and at very high temperatures. Perfect gas obeys ideal gas law and it has constant specific heats. For example if you consider air as perfect gas, Cp=1.005 kJ/kg.Is hydrogen an ideal gas?
Hydrogen gas properties can be predicted fairly accurately using the IDEAL GAS equation PV=nRT because it has very low intermolecular forces and the molecules are very small. BUT, an IDEAL gas has zero intermolecular forces and zero molecular volume so Hydrogen is not an ideal gas. Remember NOTHING IS an ideal gas.Is helium an ideal gas?
The real gas that acts most like an ideal gas is helium. This is because helium, unlike most gases, exists as a single atom, which makes the van der Waals dispersion forces as low as possible. Like a helium atom, a hydrogen molecule also has two electrons, and its intermolecular forces are small.What is an example of a real gas?
Any gas that exists is a real gas. Nitrogen, oxygen, carbon dioxide, carbon monoxide, helium etc. Real gases have small attractive and repulsive forces between particles and ideal gases do not. Real gas particles have a volume and ideal gas particles do not.What defines an ideal gas?
An ideal gas is a gas whose pressure P, volume V, and temperature T are related by the ideal gas law: PV = nRT. where n is the number of moles of the gas and R is the ideal gas constant. Ideal gases are defined as having molecules of negligible size with an average molar kinetic energy dependent only on temperature.What is difference between real and ideal gas?
Real gases have small attractive and repulsive forces between particles and ideal gases do not. Real gas particles have a volume and ideal gas particles do not. Real gas particles collide in-elastically (loses energy with collisions) and ideal gas particles collide elastically.Do ideal gases exist?
Explanation: Of course, an ideal gas does not exist except as a conceptual notion, an ideal. Physicists and chemists idealized the behaviour of real gases so that they could explain these phenomena. Under conditions of low pressure and high temperature, all gases behave like real gases, even gases such as UF6 .What are ideal and non ideal gases?
Ideal vs Non-Ideal Gases An ideal gas is one in which the molecules don't interact with each other and don't take up any space. Other gases behave much like ideal gases when they are at low pressures and temperatures. Low pressure means few interactions between gas molecules occur.What is gas behavior?
The Measureable Properties of Gases are Pressure, volume, temperature, Amount of Gas, and Density. The behavior of gases is explained through Kinetic Molecular Theory which means that all matter is made up of constantly moving atoms or molecules. 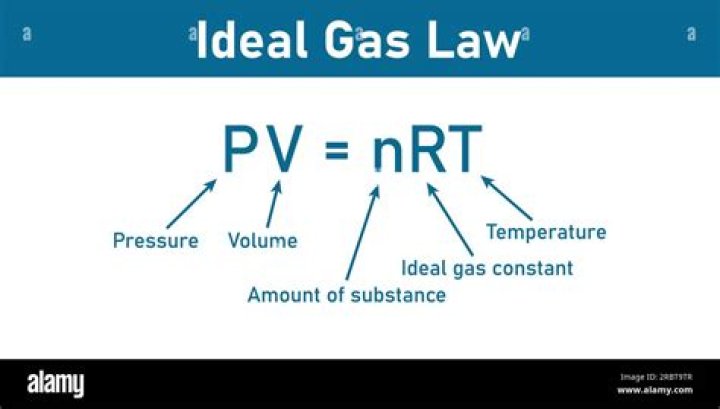
 Andrew Campbell
Andrew Campbell