How is galvanic corrosion caused?
 Isabella Bartlett
Isabella Bartlett .
People also ask, what is galvanic cell corrosion?
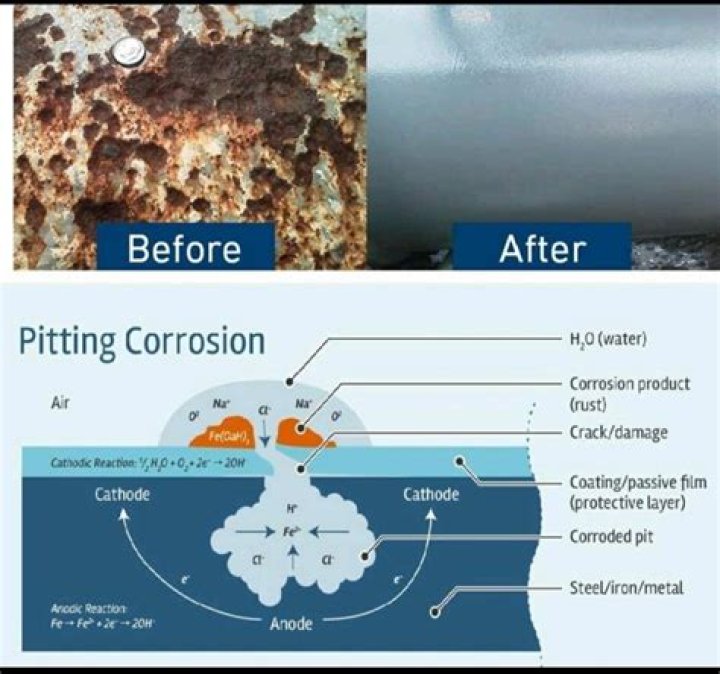
Galvanic corrosion (also called bimetalliccorrosion) is an electrochemical process in which one metalcorrodes preferentially when it is in electrical contact withanother, in the presence of an electrolyte.
Also Know, what happens during galvanic corrosion? Galvanic Corrosion. Galvanic corrosion(dissimilar-metal corrosion) is an electrochemical processin which one metal corrodes preferentially, when inelectrical contact with a different type of metal, and both metalsare immersed in an electrolyte such as water.
Just so, does galvanic corrosion require oxygen?
In the absence of dissolved oxygen or hydrogenions to maintain the cathode process, galvanic corrosiondoes not occur. It is possible to combine different metals suchas copper and steel in closed hot-water systems with littlecorrosion.
Is Zinc an anode or cathode?
By convention in standard cell notation, theanode is written on the left and the cathode iswritten on the right. So, in this cell: Zinc is theanode (solid zinc is oxidised). Silver is thecathode (silver ions are reduced).
Related Question AnswersWhat does anodic mean?
Anodic means relating to an anode. In ananodic reaction, oxidation occurs, meaning electronsare removed from the anode's surface. An anodic reaction isvery important in the corrosion of metals.Is there galvanic corrosion between stainless steel and aluminum?
Galvanic Corrosion. The combination ofaluminum and stainless steel causes galvaniccorrosion.What is anode and cathode?
Definition: The anode of a device is the terminalwhere current flows in from outside. The cathode of a deviceis the terminal where current flows out. Since electrons arenegatively charged, positive current flowing in is the same aselectrons flowing out.Which corrodes anode or cathode?
Anode - The electrode where galvanic reaction(s)generate electrons - negative ions are discharged and positive ionsare formed. Corrosion occurs at the anode. Cathode -The electrode that receives electrons - positive ions aredischarged, negative ions are formed. The cathode isprotected from corrosion.What happens when two different metals touch?
This occurs when two different metals are incontact in a corrosive or conductive environment and the currentflow changes. When two dissimilar metals are involved, thereaction is called galvanic corrosion. The further apart themetals are on the corrosive scale, the stronger the galvanicreaction between the two.Does carbon steel react with stainless steel?
So, yes, leaving stainless steel in contact withhigh carbon (non-stainless) steel can causethe stainless to corrode (although not by drawingcarbon to the surface- your informant is likely confusinggalvanic corrosion with sensitization, but that's anotherlong-winded explanation).Can I use steel rivets on aluminum?
For example, never place an aluminum rivet in alarge piece of steel. For example, when fasteningaluminum and steel in a structural application,use a fastener made of carbon steel with suitableplating.Can you put galvanized and aluminum together?
What happens when I put galvanized steeltogether with aluminum parts or put aluminumsheets on galvanized steel studs? If two metals withdifferent potentials are placed in contact and there is aconductive medium, such as sea water or condensation, therecan be a reaction, commonly known as a galvaniccell.How do you prevent crevice corrosion?
To defend against crevice corrosion in existingassets, be sure to fully drain and dry any assets exposed to wateror other solutions and avoid creating stagnant conditionsthat can spur corrosion. Also, locate existingcrevices in overlapping joints and use continuous welding orsoldering to seal the gap.How can corrosion be prevented?
Whatever your reason for wanting to stop and prevent thecorrosion of metals, here are some helpful ways to preventcorrosion of metals:- Turn to non-corrosive metals such as aluminum and stainlesssteel.
- Keep the area around the metal surface dry.
- Use drying agents and moisture barrier products.