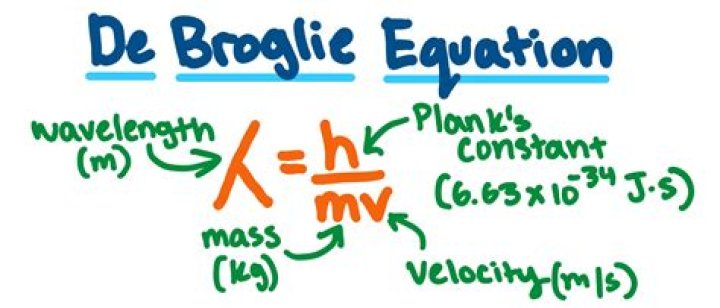
How do you use Broglie equation
 Andrew Campbell
Andrew Campbell Apply the de Broglie wave equation λ=hmv λ = h m v to solve for the wavelength of the moving electron. Step 3: Think about your result. This very small wavelength is about 1/20th of the diameter of a hydrogen atom. Looking at the equation, as the speed of the electron decreases, its wavelength increases.
What does the de Broglie equation tell us?
The de Broglie equation is an equation used to describe the wave properties of matter or particles. de Broglie suggested that particles can exhibit properties of waves, and proved that every moving particle has a matter wave associated with it. … m is the mass of a particle in kg moving at a velocity v in m/s .
What is the de Broglie wavelength used for?
According to wave-particle duality, the De Broglie wavelength is a wavelength manifested in all the objects in quantum mechanics which determines the probability density of finding the object at a given point of the configuration space.
What is the equation for the de Broglie wavelength?
The deBroglie wavelength is defined as follows: lambda = h/mv , where the greek letter lambda represents the wavelength, h is Planck’s contant, m is the particle’s mass and v is its velocity.Which of the following equations did de Broglie suggest?
πr2=nλ
What is the velocity of de Broglie wave?
Because real particles do not travel at the speed of light, De Broglie submitted velocity (v) for the speed of light (c). Through the equation λ, de Broglie substituted v/λ for ν and arrived at the final expression that relates wavelength and particle with speed.
How do you find de Broglie wavelength given mass and velocity?
Multiplying the mass and speed, we obtain the momentum of the particle: p = mv = 2.7309245*10-24 kg·m/s . If we divide the Planck constant by the momentum, we will obtain the de Broglie wavelength: h/p = 6.6261*10-34 / 2.7309245*10-24 = 2.426*10-10 m .
What is de Broglie equation explain relation between wavelength and momentum with the help of de Broglie equation?
According to de-Broglie hypothesis, the wavelength associated with moving electron of mass ′m′ is ′λe′. Using mass energy relation and Planck’s quantum theory, the wavelength associated with photon is ′λp′. If the energy (E) of electron and photon is same then relation between ′λe′ and ′λp′ is.How do you find de Broglie wavelength with kinetic energy?
If you double the kinetic energy of a particle, how does the deBroglie wavelength change? Solution: λ = h/p, E = p2/(2m), p is proportional to √E, l is proportional to 1/√E. λ2/λ1 = √(E1/E2) = 1/√2.
What is de Broglie relation?De-Broglie relation relates a body’s momentum with its wavelength. It is given as. λ=ph. where λ is its de-broglie wavelength. h is the plank’s constant.
Article first time published onWhat is the de Broglie wavelength of the electron?
A typical electron in a metal has a de Broglie wavelength is of order ~ 10 nm.
How do you find the wavelength when given kinetic energy?
To calculate the wavelength, you need to know either the frequency or the energy of the radiation. 590 kHz? E=hcλ or λ=hcE , where h is Planck’s constant.
How do you find kinetic energy given wavelength and work function?
Definition: The Maximum Kinetic Energy of a Photoelectron given Wavelength. The maximum kinetic energy of a photoelectron is given by 𝐸 = ℎ 𝑐 𝜆 − 𝑊 , m a x where ℎ is the Planck constant, 𝑐 is the speed of light, 𝜆 is the wavelength of the incident photon, and 𝑊 is the work function of the metal surface.
How do you find wavelength given kinetic energy?
Equation Number Two: λ = h/p it is here in case you migt be interested in it. Suppose an electron has momentum equal to p, then its wavelength is λ = h/p and its frequency is f = E/h.
What is the de Broglie wavelength associated with the hydrogen electron?
96×10−8 cm.
Which is not variable in Broglie equation?
Explanation: Because its value is fixed as 6.626×10^-34. Location is not included in the formula.
How do you find de Broglie wavelength with voltage?
In the case of electrons that is λde Broglie=hpe=hme⋅ve The acceleration of electrons in an electron beam gun with the acceleration voltage Va results in the corresponding de Broglie wavelength λde Broglie=hme⋅√2⋅eme⋅Va=h√2⋅me⋅e⋅Va Proof of the de Broglie hypothesis will be experimentally demonstrated with the help of …
What is the de Broglie wavelength of an electron having energy 10 Kev?
The correct option is (A) 0.12.
What is the de Broglie wavelength of electron accelerated by 100v?
Here, V=100 Volts. The de- Broglie wavelength λ is λ=1.227√Vnm.
How do you find frequency given wavelength and energy?
Wavelength is related to energy and frequency by E = hν = hc/λ, where E = energy, h = Planck’s constant, ν = frequency, c = the speed of light, and λ = wavelength.
How do I calculate kinetic energy?
In classical mechanics, kinetic energy (KE) is equal to half of an object’s mass (1/2*m) multiplied by the velocity squared. For example, if a an object with a mass of 10 kg (m = 10 kg) is moving at a velocity of 5 meters per second (v = 5 m/s), the kinetic energy is equal to 125 Joules, or (1/2 * 10 kg) * 5 m/s2.
What is the kinetic energy formula?
Kinetic energy is directly proportional to the mass of the object and to the square of its velocity: K.E. = 1/2 m v2. If the mass has units of kilograms and the velocity of meters per second, the kinetic energy has units of kilograms-meters squared per second squared.
What is the relationship between kinetic energy and frequency?
the kinetic energy of the electrons is linearly proportional to the frequency of the incident radiation above a threshold value of ν0 (no current is observed below ν0), and the kinetic energy is independent of the intensity of the radiation.
Which of the following is correct a relation between Ke and wavelength?
Phase constant = 2π/wavelength.