How do you dilute a stock solution?
 Andrew White
Andrew White .
Accordingly, how can you reduce the concentration of a solution?
Often, a worker will need to change the concentrationof a solution by changing the amount of solvent. Dilution isthe addition of solvent, which decreases theconcentration of the solute in the solution.Concentration is the removal of solvent, which increases theconcentration of the solute in thesolution.
Beside above, how do you make a dilution? Example 2: Suppose you must prepare 400 ml of adisinfectant that requires 1:8 dilution from a concentratedstock solution with water. Divide the volume needed by thedilution factor (400 ml / 8 = 50 ml) to determine the unitvolume. The dilution is then done as 50 ml concentrateddisinfectant + 350 ml water.
Similarly, you may ask, how do you dilute a 1 to 10 solution?
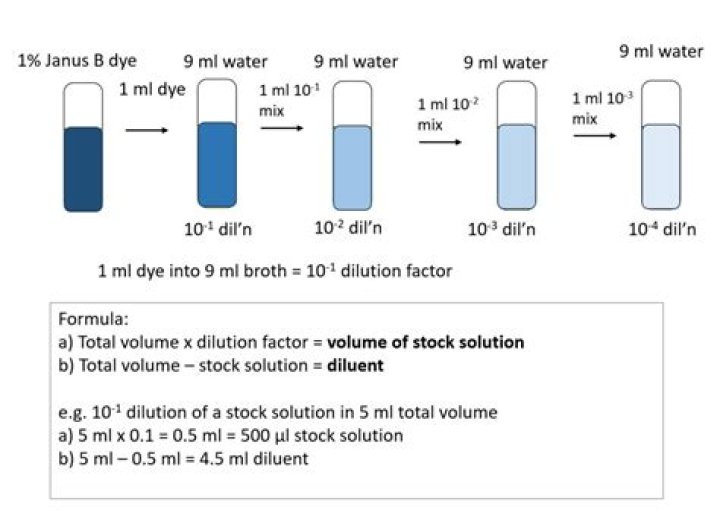
Therefore, 1:10 dilution means 1part + 9 parts of water (or other diluent). For example: if youneeded 10 mL of the 1:10 dilution, then youwould mix 1mL of the 1M NaCl with 9mL of water. Or: if you needed100mL of the 1:10 dilution, then you would mix 10mLof the 1M NaCl with 90mL of water.
How do you prepare a dilution from a stock solution?
As an example, say you need to prepare 50milliliters of a 1.0 M solution from a 2.0 M stocksolution. Your first step is to calculate the volume ofstock solution that is required. To make yoursolution, pour 25 ml of stock solution into a 50 mlvolumetric flask. Dilute it with solvent to the 50 mlline.
Related Question AnswersDoes dissolving change volume?
The new dissolves atoms still need some space inthe liquid, and most of the time this means that the volumeincreases. So the mass of the solute is now added to the solventand because the volume isn't dependant on the mass of theparticles dissolved it won't change in a way thatkeeps the density the same.What is concentration of a solution?
Concentration Definition. In chemistry,concentration refers to the amount of a substance perdefined space. Another definition is that concentration isthe ratio of solute in a solution to either solvent or totalsolution. However, the solute concentration may alsobe expressed in moles or units of volume.How do you find the concentration of a dilute solution?
You can solve for the concentration or volume ofthe concentrated or dilute solution using theequation: M1V1 = M2V2, where M1 is the concentrationin molarity (moles/Liters) of the concentrated solution, V2is the volume of the concentrated solution, M2 is theconcentration in molarity of the dilute solution(afterHow do I find the concentration of a solution?
Divide the mass of the solute by the total mass of thesolution. Set up your equation so theconcentration C = mass of the solute/total mass of thesolution. Plug in your values and solve the equationto find the concentration of your solution. In ourexample, C = (10 g)/(1,210 g) = 0.00826.What is a dilute solution?
A dilute solution is a one that has only a littlesolute dissolved in a certain amount of solvent. In other words, asmall amount of solute in between the solventmolecules.What is supersaturated solution?
Supersaturation is a solution thatcontains more of the dissolved material than could be dissolved bythe solvent under normal circumstances. It can also refer to avapor of a compound that has a higher (partial) pressure than thevapor pressure of that compound.What does c1v1 c2v2 stand for?
C1V1=C2V2 is used to calculate an unknownquantity where two solutions/mixtures are proportional …C1V1 = Concentration/amount (start) and Volume (start)C2V2 = Concentration/amount (final) and Volume (final) 1.Amounts.What is a 1 in 2 dilution?
In other words, an initial dilution is made andthen this dilution is used to make a second dilution,and so on. For example, a 1:2 serial dilutionis made using a 1 mL volume of serum. Then, 1 mL ofthis dilution is added to 1 mL of H20 furtherdiluting the sample.How do you make a 1 100 dilution?
To make a 1:100 dilution, one part of thesolution is mixed with 99 parts of additional solvent. Mixing 100µL of a stock solution with 900 µL of water makes a1:10 dilution. The final volume of the diluted sampleis 1000 µL (1 mL) and the concentration is 1/10 that of theoriginal solution.How do I make a 10% solution?
We can make 10 percent solution by volumeor by mass. A 10% of NaCl solution by mass has tengrams of sodium chloride dissolved in 100 ml of solution.Weigh 10g of sodium chloride. Pour it into a graduated cylinder orvolumetric flask containing about 80ml of water.What is a 1/4 dilution?
A 1:4 dilution ratio means that a simpledilution contains one part concentrated solution or soluteand four parts of the solvent, which is usually water. For example,frozen juice that requires one can of frozen juice plus four cansof water is a 1:4 simple dilution.How do you make a 1 200 dilution?
1 ml of serum + 199 ml of diluent =1/200 dilution. 1 ml of 1/200dilution + 49 ml of diluent = 1/50. The first step inmaking a serial dilution is to take a known volume(usually 1ml) of stock and place it into a known volume ofdistilled water (usually 9ml). This produces 10ml of the dilutesolution.What is a 1% solution?
A one percent solution is defined as1 gram of solute per 100 milliliters final volume. Forexample, 1 gram of sodium chloride, brought to a finalvolume of 100 ml with distilled water, is a 1% NaClsolution. To help recall the definition of a 1%solution, remember that one gram is the mass ofone milliliter of water.What is a 1/3 dilution?
In medicine and chemistiry, dilulion 1:3 meansdilute one part concentrate with solvent, such that the finalvolume is 3 part. In some photography formularies, however,"dilution 1:3" means. dilute one part of concentrate with 3part of water.What is a 1 in 20 dilution?
These two components proportionally combine to create adilution. For example, a 1:20 dilutionconverts to a 1/20 dilution factor. Multiply thefinal desired volume by the dilution factor to determine theneeded volume of the stock solution. In our example, 30 mL x1 ÷ 20 = 1.5 mL of stocksolution.How do you make a 1 acetic acid solution?
To make a 1 M solution acetic acid,dissolve 60.05 g acetic acid in 500 mL distilled ordeionized water in a 1000-mL volumetric flask. Since aceticacid is a liquid, the acid may also be measured byvolume. Divide the mass of acid by its density (1.049 g/mL)to determine the volume (57.24 mL).How do you make a percent solution?
To calculate the mass percent or weightpercent of a solution, you must divide the mass ofthe solute by the mass of the solution (both the solute andthe solvent together) and then multiply by 100 to change it intopercent. Your workbook has some examples of calculationsinvolving weight percent in example 1.How do you make a solid solution?
Making Solutions from Pure Solids- First, weigh out the correct mass of solute.
- Dissolve the solute in water, keeping the volume less than thedesired total volume of solution.
- Dilute the solution to the desired total volume ofsolution.





