A decrease in the number of moles on the product side means lower entropy. An increase in the number of moles on the product side means higher entropy. If the reaction involves multiple phases, the production of a gas typically increases the entropy much more than any increase in moles of a liquid or solid..
Similarly one may ask, how do you know if entropy is increasing?
Entropy increases as you go from solid to liquid to gas, and you can predict whether entropy change is positive or negative by looking at the phases of the reactants and products. Whenever there is an increase in gas moles, entropy will increase.
Secondly, what would cause entropy to increase in a reaction? Entropy also increases when solid reactants form liquid products. Entropy increases when a substance is broken up into multiple parts. The process of dissolving increases entropy because the solute particles become separated from one another when a solution is formed. Entropy increases as temperature increases.
Likewise, people ask, which are examples of entropy increasing?
A campfire is an example of entropy. The solid wood burns and becomes ash, smoke and gases, all of which spread energy outwards more easily than the solid fuel. Ice melting, salt or sugar dissolving, making popcorn and boiling water for tea are processes with increasing entropy in your kitchen.
Is more entropy positive or negative?
The reaction will occur, as in an exothermic reaction H is negative, and if the entropy increases, then S is positive, so: Total entropy change is positive, so reaction is feasible. The reaction can never occur, as H is positive and S is negative: The total entropy change is negative and so the reaction cannot occur.
Related Question Answers
Does boiling water increase or decrease entropy?
The entropy increases whenever heat flows from a hot object to a cold object. It increases when ice melts, water is heated, water boils, water evaporates. The entropy increases when a gas flows from a container under high pressure into a region of lower pressure.What does entropy depend on?
Energy and Entropy Entropy is a measure of the random activity in a system. The entropy of a system depends on your observations at one moment. How the system gets to that point doesn't matter at all. If it took a billion years and a million different reactions doesn't matter.What does a negative entropy mean?
Entropy is the amount of disorder in a system. Negative entropy means that something is becoming less disordered. In order for something to become less disordered, energy must be used. The second law of thermodynamics states that the world as a whole is always in a state of positive entropy.What is the equation for entropy?
If the process is reversible, then the change in entropy is equal to the heat absorbed divided by the temperature of the reversible process. In the equation, Q is the heat absorbed, T is the temperature, and S is the entropy. Entropy is also the measure of energy not available to do work for your system.Which process is an example of entropy decreasing?
Evolution (animals and plants) are an example of entropy of a local system decreasing. However the sun input energy into the system, and therefore entropy increased all over other sections of the universe while this small local entropy decrease was occurring on earth.Is zero entropy possible?
The Basic Law The third law of thermodynamics states that the entropy of a system approaches a constant value as the temperature approaches zero. The entropy of a system at absolute zero is typically zero, and in all cases is determined only by the number of different ground states it has.What increases and decreases entropy?
Does the entropy increase or decrease for the following reactions? Answers: INCREASES - The number of particles in the system increases, i.e. one particle decomposes into two. DECREASES - The number of particles in the system decreases, i.e. there are four moles of gas reactants and only 2 moles of gas products.What is the unit of entropy?
The SI unit for Entropy (S) is Joules per Kelvin (J/K). A more positive value of entropy means a reaction is more likely to happen spontaneously.How is entropy generated?
Entropy production (or generation) is the amount of entropy which is produced in any irreversible processes such as heat and mass transfer processes including motion of bodies, heat exchange, fluid flow, substances expanding or mixing, anaelastic deformation of solids, and any irreversible thermodynamic cycle,Why is entropy important?
Entropy is a bookkeeping device, which tells us about the flow and distribution of energy. For any process to occur to occur spontaneously, it is a necessary condition that the entropy of the system undergoing the process should increase. If the entropy decreases, then that process cannot occur spontaneously.When can entropy be negative?
A negative change in entropy indicates that the disorder of an isolated system has decreased. For example, the reaction by which liquid water freezes into ice represents an isolated decrease in entropy because liquid particles are more disordered than solid particles.Why Does entropy increase over time?
Entropy (arrow of time) As one goes "forward" in time, the second law of thermodynamics says, the entropy of an isolated system can increase, but not decrease. Hence, from one perspective, entropy measurement is a way of distinguishing the past from the future.What are the three laws of thermodynamics?
The three laws of thermodynamics define physical quantities (temperature, energy, and entropy) that characterize thermodynamic systems at thermodynamic equilibrium. The laws describe how these quantities behave under various circumstances, and preclude the possibility of certain phenomena (such as perpetual motion).What does it mean when entropy is positive?
Enthalpy, Entropy, 2nd Law, and 3rd Law. Entropy, S, is a state function and is a measure of disorder or randomness. A positive (+) entropy change means an increase in disorder. The universe tends toward increased entropy. All spontaneous change occurs with an increase in entropy of the universe.What is enthalpy and entropy?
Enthalpy is a measure of heat content of the system, whereas entropy is the measure of change in (enthalpy/temperature). Enthalpy is a measure of heat content of the system, whereas entropy is the measure of change in (enthalpy/temperature).Does pressure affect entropy?
The entropy of a system decreases with an increase in pressure. Entropy is a measure of how much the energy of atoms and molecules become more spread out in a process. If we increase the pressure on the system, the volume decreases. The energies of the particles are in a smaller space, so they are less spread out.Is entropy a force?
In physics, an entropic force acting in a system is an emergent phenomenon resulting from the entire system's statistical tendency to increase its entropy, rather than from a particular underlying force on the atomic scale. It should not be referred to as a distribution of the entropy in the space.What is entropy in simple words?
The entropy of an object is a measure of the amount of energy which is unavailable to do work. Entropy is also a measure of the number of possible arrangements the atoms in a system can have. In this sense, entropy is a measure of uncertainty or randomness.What is enthalpy in simple terms?
Enthalpy. When a substance changes at constant pressure, enthalpy tells how much heat and work was added or removed from the substance. Enthalpy is similar to energy, but not the same. When a substance grows or shrinks, energy is used up or released. 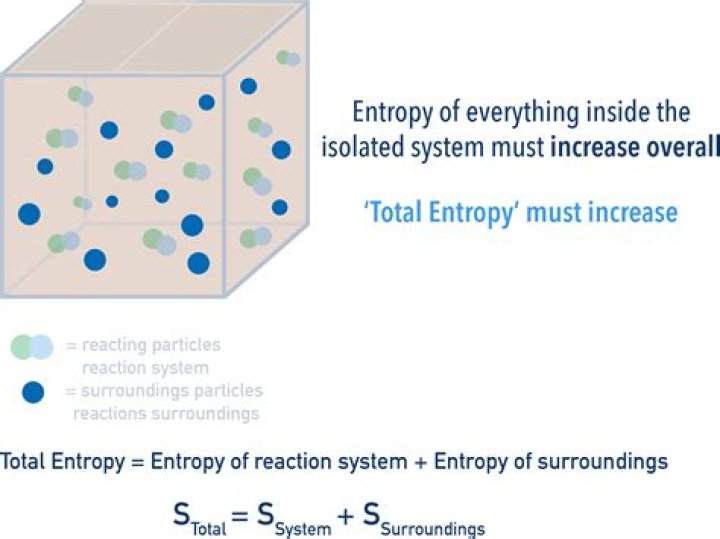
 John Peck
John Peck 




