If you have a solution, you multiply the molarity by the volume in litres. There are two steps: Multiply the volume by the density to get the mass. Divide the mass by the molar mass to get the number of moles..
In respect to this, how do you convert to molarity?
To convert from % solution to molarity, multiply the % solution by 10 to express the percent solution grams/L, then divide by the formula weight.
One may also ask, how do you find moles from volume and molarity? Compute the volume of a solution in liters, given the number of moles and molarity, by dividing the number of moles by the molarity in units of moles per liter. For example, a solution containing 6.0 moles and a having a molarity of 3.0 moles per liter has a volume of 2.0 moles per liter.
Also to know is, how do you convert molarity to Grams?
You can convert molarity into grams per liter by multiplying it with the molecular (Molar) mass, i.e. the number of grams/Mol. Molarity is Mol/L which when multiplied with the Molecular Mass (sometimes also called Molecular Weight) becomes grams/liter.
What is Molality formula?
The formula for molality is m = moles of solute / kilograms of solvent. In problem solving involving molality, we sometimes need to use additional formulas to get to the final answer. One formula we need to be aware of is the formula for density, which is d = m / v, where d is density, m is mass and v is volume.
Related Question Answers
What is the molarity of 5% HCl?
Dilutions to Make a 1 Molar Solution
| Concentrated Reagents | Density | Molarity (M) |
| Hydrochloric acid 36% | 1.18 | 11.65 |
| Hydrochloric acid 32% | 1.16 | 10.2 |
| Hydrofluoric acid 40% | 1.13 | 22.6 |
| Nitric acid 70% | 1.42 | 15.8 |
How do you find the molarity of NaOH?
Method 1: - Calculate the number of moles of solute present. mol NaOH. = 15.0g NaOH. x. 1 mol NaOH. 40.0 g NaOH. mol NaOH.
- Calculate the number of liters of solution present. L soln. = 225 mL. x. 1 L. =
- Divide the number of moles of solute by the number of liters of solution. M. = 0.375 mol NaOH. = 1.67 M NaOH. 0.225 L soln.
How many grams are a liter?
How many grams in 1 liter? The answer is 1000. We assume you are converting between gram [water] and liter. You can view more details on each measurement unit: grams or liter The SI derived unit for volume is the cubic meter.What is the molarity of NaCl?
The molar concentration of NaCl in the solution is therefore: c(NaCl) = 11.6 g58 g/mol / 104.3 mL = 0.00192 mol/mL = 1.92 mol/L. Here, 58 g/mol is the molar mass of NaCl.What does W W mean?
w/w. (chemistry) "weight for weight" or "weight by weight", the proportion of a particular substance within a mixture, as measured by weight or mass.How do you find molarity of HCl?
Ultimately, to find the maximum molarity of the HCl in water, we must use the concentration equation for molarity, which is defined as moles of the solute divided by the liters of the solution (M = moles of solute/ liters of solution).How do you convert percentage to moles?
Divide the molecular weight into the weight of solute in the solution to find the number of moles: 2.7 g ÷ 58 g/mole = 0.047 moles. Divide by the volume of the solution to find molarity: M = (0.047 moles ÷ 0.09 liters) = 0.52 moles/liter.How many moles are in a milliliter?
How many mole/litre in 1 mol/mL? The answer is 1000. We assume you are converting between mole/litre and mole/milliliter. You can view more details on each measurement unit: mole/litre or mol/mL The SI derived unit for amount-of-substance concentration is the mole/cubic meter.How many moles are in a liter?
How many Liters would each mole take up at these temperatures?! As long as the gas is ideal, 1 mole = 22.4L.Is g/l the same as molarity?
Molarity refers to the molar concentration of a solution, that is, the number of moles of solute dissolved in 1 liter of solution, as mol/L, abbreviated as M. The molar mass of NaCl is 58.44276 g/mol.How many ml is a gram?
How many grams in 1 milliliters? The answer is 1. We assume you are converting between gram [water] and milliliter. You can view more details on each measurement unit: grams or milliliters The SI derived unit for volume is the cubic meter.What is a Millimolar?
Chemists use molarity as a measure of concentration of a particular compound in solution. They define molarity as the number of moles per liter. This is equivalent to a 1,000 Mmol (millimolar, also sometimes written as mM) solution, which is one that contains 1,000 Mmol per liter.How many grams of NaOH are in 500mL of a 1m solution?
40 grams
What does moles stand for?
The mole (symbol: mol) is the unit of measurement for amount of substance in the International System of Units (SI). It is defined as exactly 6.02214076×1023 constitutive particles, which may be atoms, molecules, ions, or electrons.What is M in chemistry?
Molarity (M) indicates the number of moles of solute per liter of solution (moles/Liter) and is one of the most common units used to measure the concentration of a solution. Molarity can be used to calculate the volume of solvent or the amount of solute.How do you make a 1 mM solution?
A 1M solution would consist of 342.3 grams sucrose in one liter final volume. A concentration of 70 mM is the same as 0.07 moles per liter. Take 0.07 moles/liter times 342.3 grams per mole and you have 23.96 grams needed per liter. To make 200 milliliters of your solution multiply grams/liter by liters needed.How do you convert volume to moles?
Converting from moles to volume (liters): Multiply your mole value by the molar volume constant, 22.4L. Converting from particles (atoms, molecules, or formula units) to moles: Divide your particle value by Avogadro's number, 6.02×1023. Remember to use parentheses on your calculator!What is the formula for molarity of a solution?
To calculate molarity, divide the number of moles of solute by the volume of the solution in liters. If you don't know the number of moles of solute but you know the mass, start by finding the molar mass of the solute, which is equal to all of the molar masses of each element in the solution added together.How many moles are in molarity?
The M is the symbol for molarity, the mol/L is the unit used in calculations. Example #2: Suppose you had 2.00 moles of solute dissolved into 1.00 L of solution. 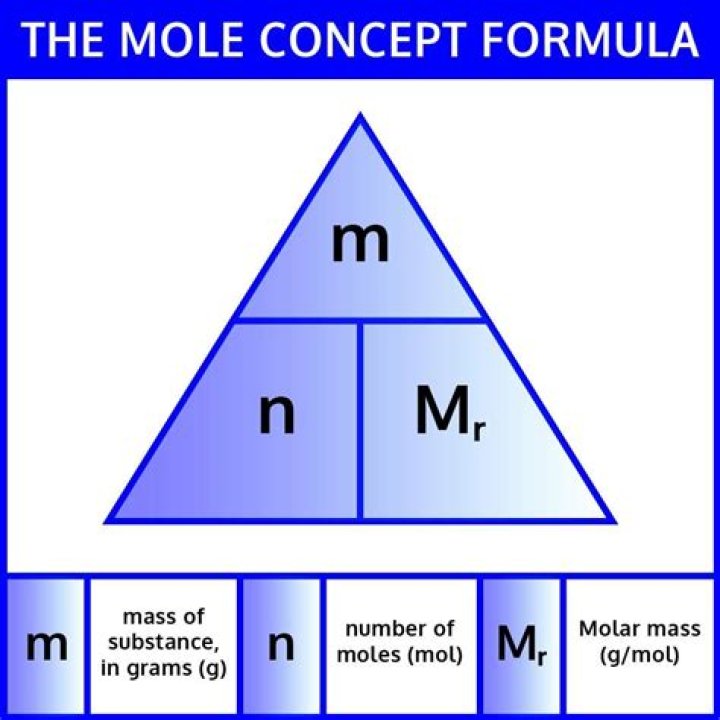
 Emma Terry
Emma Terry