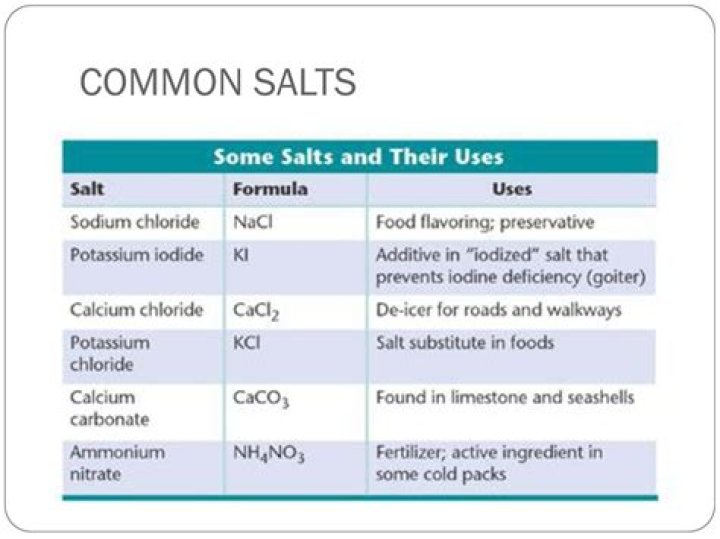
How do we name salts
 Isabella Bartlett
Isabella Bartlett The name of a salt starts with the name of the cation (e.g., sodium or ammonium) followed by the name of the anion (e.g., chloride or acetate). Salts are often referred to only by the name of the cation (e.g., sodium salt or ammonium salt) or by the name of the anion (e.g., chloride salt or acetate salt).
How do salts get their names?
The name of a salt has two parts. The first part comes from the metal, metal oxide or metal carbonate. The second part comes from the acid. … hydrochloric acid always produces salts that end in chloride and contain the chloride ion, Cl.
What are the salt names?
Name of the saltChemical formulaNegative ionSodium chlorideNaClCl−Magnesium sulphateMgSO4SO2−4Calcium carbonateCaCO3
What is the rule of naming salts?
Salts are named by listing the names of their component ions, cation first, then anion.How do we name a salt give two examples?
Naming Salts (Ionic Compounds) Saltsare ionic compounds which, when dissolved in water, break up completely into ions. They arise by thereaction of acids with bases, and they always contain either a metal cation or a cation derived from ammonium (NH4+). Examples of salts include NaCl, NH4F, MgCO3, and Fe2(HPO4)3.
How do you name a binary salt?
Compounds formed from metal ions and simple nonmetal ions are called binary salts. LiCl is the formula of a typical binary salt. To write the chemical name for it we will start with the names of the two elements in the salt. Type the name of the element Li and then press Enter.
How do you name salts acids and bases?
- Acid + metal oxide → salt + water.
- For example:
- Sulfuric acid + copper oxide → copper sulfate + water.
- Alkalis are soluble bases. …
- Acid + alkali → salt + water.
- For example:
- Nitric acid + sodium hydroxide → sodium nitrate + water.
How many salts are there in chemistry?
The different types of salts are: normal salt, acid salt, basic salt and double salt. 1.How do you identify a salt in chemistry?
- Look at the Appearance of the Compound.
- Check the Effect of Heating.
- Conduct a Flame Test.
- Test Its Reaction with Hydrochloric Acid.
- Note Its Solubility in Water.
- Sieve analysis.
- Moisture analysis.
- Add some dilute hydrochloric acid to a beaker.
- Add powdered zinc carbonate to some acid, one spatula at a time, stirring to mix. …
- Continue adding powder until it is in excess (some unreacted powder is left over).
- Filter the mixture in the beaker to remove the excess zinc carbonate.
What is salts Name any two salts?
The names and formulae of two salts are sodium chloride, NaCl, and ammonium chloride, NH4Cl, respectively. Sodium chloride salt (NaCl) is formed from an acid, hydrochloric acid (HCl), and a base (NaOH).
What is salt name any salt and give the formula?
a A salt is a compound formed from an acid by the replacement of the hydrogen in the acid by a metal. Example: Sodium chloride – NaCl; It is obtained from hydrochloric acid and sodium metal. Ammonium chloride – NH4Cl; It is obtained from ammonia and hydrochloric acid.
What is salt give the name and formula?
Sodium chloride, also known as table salt, is an ionic compound with the chemical formula NaCl, representing a 1:1 ratio of sodium and chloride ions. It is commonly used as a condiment and food preservative. Salt can be created by adding two very reactive elements together: sodium (Na(s) metal and chlorine (Cl2(g) gas.
How do you name acids in chemistry?
Acids are named based on their anion — the ion attached to the hydrogen. In simple binary acids, one ion is attached to hydrogen. Names for such acids consist of the prefix “hydro-“, the first syllable of the anion, and the suffix “-ic”. Complex acid compounds have oxygen in them.
How are bases named?
How are bases named? Bases are named in the same way as other ionic compounds-the name of the cation is followed by the name of the anion.
What is ternary salt?
A ternary salt is a compound containing three different elements. In a binary compound, the first name will be a metal ion. … A ternary acid is an acid that has the elements hydrogen and oxygen along with another element, often a nonmetal. Or, they contain hydrogen in addition to a polyatomic ion.
Is NaCl a binary salt?
In a binary compound, there may be only one of each element. We see this with sodium chloride (salt) NaCl, which has one sodium (Na) and one chlorine (Cl). … From acids to salts, binary compounds are always nearby.
What is a binary salt definition?
Binary salt (Chem.), a salt of the oxy type conveniently regarded as composed of two ingredients (analogously to a haloid salt), viz., a metal and an acid radical.
How are salts made?
salt, in chemistry, substance produced by the reaction of an acid with a base. A salt consists of the positive ion (cation) of a base and the negative ion (anion) of an acid. The reaction between an acid and a base is called a neutralization reaction.
Are all salts salty?
Not all salts are safe to eat, and not all of them taste salty. The cation determines if a salt has a salty flavour, and the anion determines the intensity of that flavour. To interact with our taste receptors, salts first have to split back – or dissociate – into their ions.
What is salt analysis?
Salt analysis is the quantitative and qualitative analysis of cations and anions present in a given inorganic salt. The salt analysis involves a series of steps that help in identifying a salt and its constituent ions. … Cations can be classified into six different groups according to the properties that they possess.
What are 5 examples of salts?
Various salts can elicit all 5 basic tastes, for example, sweet (lead diacetate, which causes lead poisoning when ingested), salty (sodium chloride), bitter (magnesium sulfate), sour (potassium bitartrate), and savoury (monosodium glutamate) or umami.
What are the 10 examples of salts?
- Sodium Chloride or Common Salt (NaCl)
- Sodium Hydroxide (NaOH)
- Sodium Carbonate or Washing Soda (Na2CO3.10H2O)
- Baking Soda or Sodium Bi-carbonate (NaHCO3)
- Bleaching Powder or Calcium Hypochlorite.
- Plaster of Paris or Hemihydrate Calcium sulphate, CaSO4 1/2 H2O.
What are the 5 salts?
Today, we are going to focus on five of the most commonly used and talked about salts: refined table salt, sea salt, kosher salt, pink Himalayan salt, and Celtic sea salt.
What are the salts in chemistry?
In chemical terms, salts are ionic compounds. To most people, salt refers to table salt, which is sodium chloride. Sodium chloride forms from the ionic bonding of sodium ions and chloride ions. There is one sodium cation (Na+) for every chloride anion (Cl–), so the chemical formula is NaCl (Fig.
How are salts formed quizlet?
A: Salts are chemical compounds that are usually formed from the combination of an acid and a base in water. … Q: An example of combining an acid and a base to form a salt is combining a water solution of hydrochloric acid (HCl) with a water solution of sodium hydroxide base (NaOH).
What do we call salts that dissolve in water?
Ionic compounds such as sodium chloride, that dissolve in water and dissociate to form ions, are called electrolytes.
What is a salt Name any two salts Class 7?
BaseAcidSaltSodium hydroxide NaOHCarbonic acid H2CO3Sodium carbonate Na2CO3
What is a salt Class 10?
Salts: Salts are the ionic compounds which are produced after the neutralization reaction between acid and base. Salts are electrically neutral. There are number of salts but sodium chloride is the most common among them. Sodium chloride is also known as table salt or common salt.
How are salts Named give two examples class 7th?
The salts are named after the metal supplied by the base and the radical supplied by the acid. Example 1: Sodium chloride (common salt) is formed from hydrochloric acid and sodium hydroxide. … Example 2: Ammonium sulphate (salt) is formed from sulphuric acid and ammonium hydroxide.
What is salts formula?
Pure table salt is sodium chloride, with the formula NaCl. Equal numbers of sodium and chlorine atoms are arranged in a crystal ionic lattice.