How can you measure the plant pieces to determine the rate of osmosis
 Emily Sparks
Emily Sparks The rate of osmosis is determined by the total number of particles dissolved in the solution. The more particles dissolved, the faster the rate of osmosis. If a membrane is present, water will flow to the area with the highest concentration of solute.
How do you measure the rate of osmosis?
The rate of osmosis is determined by the total number of particles dissolved in the solution. The more particles dissolved, the faster the rate of osmosis. If a membrane is present, water will flow to the area with the highest concentration of solute.
What factors determine rate and direction of osmosis?
Concentration gradient – The movement of osmosis is affected by the concentration gradient; the lower the concentration of the solute within a solvent, the faster osmosis will occur in that solvent. Light and dark – They are also factors of osmosis; since the brighter the light, the faster osmosis takes place.
How do you test for osmosis in plant cells?
Another way of looking at osmosis in plant cells is to mount a piece of onion skin, or beetroot on microscope slides in drops of different concentrations of sugar or salt. Observe the cells for a few minutes. It is easy to see the process of plasmolysis in beetroot because the cell sap is red.How do you identify osmosis?
Blisters can vary from small pinhead blisters, to areas as large as the palm of a hand. The presence of any fluid behind a blister indicates a potential problem. If the fluid has a pungent, vinegary odor or feels greasy or sticky when rubbed between the thumb and forefinger, there is a high probability of osmosis.
How would you calculate the water potential in the plant cells?
The formula for calculating water potential is Ψ = ΨS + ΨP. Osmotic potential is directly proportional to the solute concentration. If the solute concentration of a solution increases, the potential for the water in that solution to undergo osmosis decreases.
What does the rate of osmosis mean?
The uptake of water by plant cells or loss of water from them is due to osmosis. Water passes from a more dilute solution to a more concentrated solution through the partially permeable cell membrane. … The more dilute a solution, the higher its water potential.
How does salt concentration affect osmosis in plant cells?
Salt triggers osmosis by attracting the water and causing it to move toward it, across the membrane. … If the concentration of salt inside a cell is the same as the concentration of salt outside the cell, the water level will stay the same, creating an isotonic solution.How does osmosis differ in plant and animal cells?
Osmosis affects plant and animal cells differently because plant and animal cells can tolerate different concentrations of water. … However, plant cells need more water than animal cells, and will not burst in a hypotonic solution due to their thick cell walls; hypotonic solutions are ideal for plant cells.
What does osmosis rate depend on?The rate of osmosis always depends on the concentration of solute. The process is illustrated by comparing an environmental or external solution to the internal concentration found in the body.
Article first time published onWhat factors determine the rate and direction of osmosis quizlet?
the concentration of water determines the direction in which molecules will move during osmosis. the point at which water molecules enter and leave a cell at the same rate. the concentration inside and outside of the cell are equal.
What factors affect the rate of osmotic movement of water?
The rate of osmosis varies with a number of factors, including temperature, pressure, and the difference in solute concentrations between two solutions separated by a selectively permeable membrane.
Does the rate of osmosis increase with concentration?
The concentration of a solute affects the rate of osmosis over time, in a way where, the higher the concentration of a solute, the faster the rate of osmosis. This happens because, in a semi-permeable membrane the water is the only through that can move through. … That results in the rate of osmosis being faster.
How do you remember osmosis?
An Australian woman, Oz for short, only mows (osmosis) her lawn in the rain (water). When she does so, the grass changes from being very high to being very low.
What are 3 examples of osmosis?
- Fish Absorb Water Through Their Skin and Gills.
- Red Blood Cells Placed Into Freshwater. …
- Salt on Slugs. …
- Plants Absorb Water From The Soil. …
- Potato In Sugar Solution. …
- Raisin In Water. …
How do plants use osmosis?
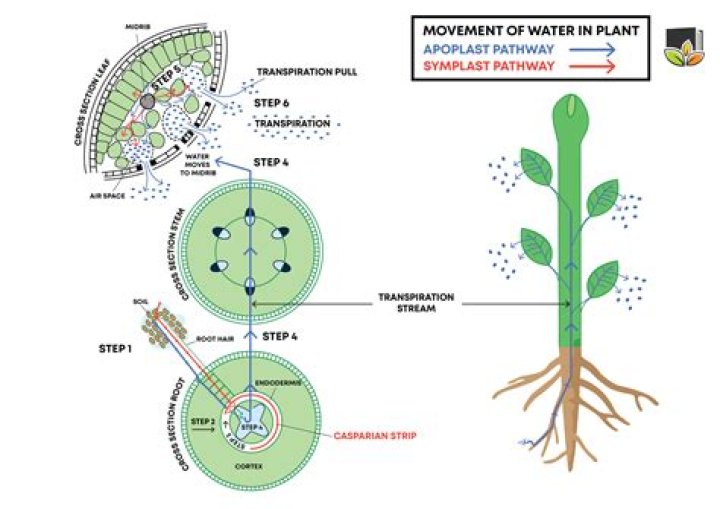
In plants, water enters the root cells by osmosis, and moves into tubes called xylem vessels to be transported to the leaves. … When water evaporates from the leaves (through tiny pores called stomata), more water is drawn up from the root xylem cells to replace that which has been lost.
How do you measure leaf water potential?
Leaf water potential measurements are easily and accurately obtained using the chilled-mirror dew point technique of the WP4C. The recommended procedure involves the abrasion of the leaf cuticle to speed equilibration.
What does water potential measure?
Water potential is a measure of the potential energy in water, or the difference in potential energy between a given water sample and pure water (at atmospheric pressure and ambient temperature).
How do you calculate pressure potential?
Divide the depth in meters measured in Step 1 by 10 and add one to the result to calculate the amount of atmospheres of pressure present at the depth. Multiply your result from Step 2 by the difference in depth measured in Step 1 to get the pressure potential per unit volume of the water at the depth you are measuring.
How does osmosis keep plant stems rigid?
Osmosis keeps plant stems rigid by way of the large vacuole. When abundant water is taken in by the plant, the large central vacuole (a large empty…
Why is osmosis important in plant cells?
Osmosis is responsible for the ability of plant roots to draw water from the soil. Plants concentrate solutes in their root cells by active transport, and water enters the roots by osmosis. Osmosis is also responsible for controlling the movement of guard cells.
What is an example of osmosis in plants?
In the plant cells Another classic example of osmosis in plants is the swelling up and shrinking of potato cells when slices of potato are dipped in a hypotonic solution and hypertonic solutions respectively.
How does salt water affect plants?
When the salt concentrations in the soil are higher than inside the root cells, the soil will draw water from the root, and the plant will wilt and die. This is the basic way in which salinization affects plant production. … When combined with irrigation and poor drainage it can lead to permanent soil fertility loss.
How does salt water affect plant cells?
If you water a plant with salt water, it will wilt, and will eventually die. This is due to the fact that the salt water is a hypertonic solution when compared to the plant cells, and water inside the plant cells will diffuse by osmosis out of the cells in order to reduce the concentration of the salt solution.
What happens to plant cell in salt water?
When plant cells are put in really salty water, water diffuses/moves out of the cell and the central vacuole shrinks. When animal cells are put in salty water, water diffuses/moves out of the cell and the cell shrivels up.
How would you demonstrate the process of osmosis using a potato explain with a suitable diagram?
- Take a potato tuber, remove its outer covering from one end and cut the same end flat. …
- Scoop out a cavity from the other end of the tuber running almost upto the bottom.
- Fill the cavity with the sugar solution and fit an airtight cork fitted with a capillary tube on the upper end of the cavity (fig.
How do you explain osmosis to a child?
Osmosis allows water to pass through cell membranes. The transfer of water from a dilute solution to a concentrated solution over a partly permeable membrane is defined as osmosis.
How do you compare tonicity?
If a solution has a higher concentration of solutes (less water) than another it is said to be hypertonic. A hypotonic solution has a lower concentration of solutes and more water than another solution. Isotonic solutions contain the same concentration of solutes.
What is osmosis which factor affects osmosis in a cell?
Osmosis is movement of only water molecules through a semi permeable membrane from their region of higher concentration to their region of lower concentration. The factors that affect the rate of osmosis are concentration of cell sap, the flow time rate and temperature.
What is tonicity in osmosis?
The ability of an extracellular solution to make water move into or out of a cell by osmosis is known as its tonicity. Tonicity is a bit different from osmolarity because it takes into account both relative solute concentrations and the cell membrane’s permeability to those solutes.
What determines the direction of water movement into or out?
Generally, these factors are based on two things: hydrostatic pressure and osmotic pressure. Hydrostatic pressure would cause water to flow from an area of high pressure to an area of low pressure.