Does SDS disrupt quaternary structure
 Emily Sparks
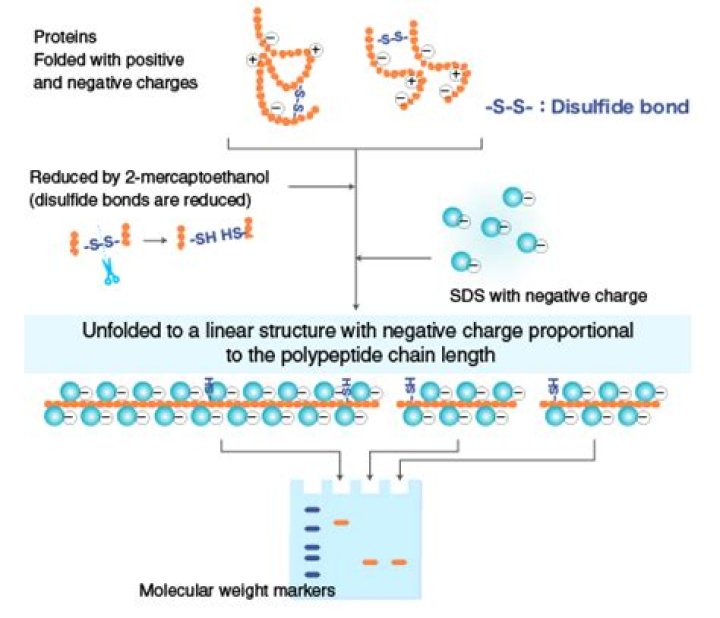
Emily Sparks SDS disrupts the secondary, tertiary and quaternary structure of the protein to produce a linear polypeptide chain coated with negatively charged SDS molecules. … try to obtain also molecular weights markers both for non-denaturing and SDS PAGE separately.
Does SDS break quaternary structure?
If you have a protein with quaternary structure, SDS-PAGE will give you the MW of the polypeptide subunits, since the SDS will denature the protein and so dissociate it into its subunits.
How does SDS affect protein structure?
SDS is an amphipathic surfactant. It denatures proteins by binding to the protein chain with its hydrocarbon tail, exposing normally buried regions and coating the protein chain with surfactant molecules. … For this reason, separation on a polyacrylamide gel in the presence of SDS occurs by mass alone.
What affects the quaternary structure of a protein?
Quaternary structure is held together by noncovalent bonds between complementary surface hydrophobic and hydrophilic regions on the polypeptide subunits. Additionally, acidic and basic side chains can form salt linkages.Does SDS disrupt tertiary structure?
in How SDS-PAGE Works. SDS is a detergent that is present in the SDS-PAGE sample buffer where, along with a bit of boiling, and a reducing agent (normally DTT or β-ME to break down protein–protein disulfide bonds), it disrupts the tertiary structure of proteins. This brings the folded proteins down to linear molecules.
What causes quaternary structure?
Quaternary Structure: Protein Chains Combine to Make Protein Complexes. Secondary and tertiary structures are determined by a protein’s sequence of amino acids, or primary structure. … Some proteins are made up of more than one amino acid chain, giving them a quaternary structure.
Does SDS disrupt secondary structure?
When SDS meets up with your protein, SDS’s hydrocarbon tail dissolves any hydrophobic region of the protein, while the sulfate end breaks non-covalent ionic bonds. This causes your protein to lose its secondary and tertiary structure, and well… unfold.
What stabilizes quaternary protein structure?
The quaternary structure of macromolecules is stabilized by the same non-covalent interactions and disulfide bonds as the tertiary structure, and can also be affected by formulation conditions.What bonds interactions hold quaternary structure together?
The quaternary structure of a protein is the association of several protein chains or subunits into a closely packed arrangement. … The subunits are held together by hydrogen bonds and van der Waals forces between nonpolar side chains.
How does SDS help disrupt the cell membrane?Denaturing detergents can be anionic such as sodium dodecyl sulfate (SDS) or cationic such as ethyl trimethyl ammonium bromide. These detergents totally disrupt membranes and denature proteins by breaking protein-protein interactions.
Article first time published onDoes SDS-PAGE break disulfide bonds?
Sodium dodecyl sulfate (SDS) is an anionic detergent used to denature proteins prior to gel electrophoresis. … However, SDS does not break down any of the disulfide bonds that participate in many tertiary structures; treatment with DTT, described below, is often necessary to break down disulfide bonds.
Does SDS-PAGE break covalent bonds?
SDS is a detergent composed of a hydrophobic hydrocarbon tail attached to an ionic sulphate group and a key component of loading buffer,. When SDS meets up with your protein, SDS’s hydrocarbon tail dissolves any hydrophobic region of the protein, while the sulfate end breaks non-covalent ionic bonds.
Does SDS PAGE denature proteins?
The addition of SDS to the protein denatures the proteins and covers them in a uniformly-distributed, net negative charge. This allows the migration of proteins towards the positive electrode during electrophoresis.
Does SDS PAGE separate amino acids?
SDS-PAGE is a method of separating proteins based on their molecular mass. SDS (sodium dodecyl sulfate) is a detergent that binds proteins and covers them with a negative charge. In general, one SDS molecule binds to two amino acids.
Which factors affect the migration of protein in SDS PAGE?
The higher the negative charge density (more charges per molecule mass), the faster a protein will migrate. At the same time, the frictional force of the gel matrix creates a sieving effect, regulating the movement of proteins according to their size and three-dimensional shape.
What does it mean to denature a protein and why is this important for SDS PAGE?
Since we are trying to separate many different protein molecules of different shapes and sizes, we first want them denatured so that the proteins no longer have any secondary, tertiary or quaternary structure (i.e. we want them to retain only their primary amino acid structure).
Why do smaller molecules move faster in SDS PAGE?
When proteins are separated by electrophoresis through a gel matrix, smaller proteins migrate faster due to less resistance from the gel matrix. Other influences on the rate of migration through the gel matrix include the structure and charge of the proteins.
What is the SDS function in the SDS PAGE gel?
SDS (sodium dodecyl sulfate) is an anionic detergent that unfolds and denatures proteins, coating proteins in negative charge. It is added in excess to the proteins, so that the proteins’ intrinsic charge is covered, and a similar charge-to-mass ratio is obtained for all proteins.
What interactions are responsible for maintaining quaternary protein structure select all that apply?
Quaternary structure describes how polypeptide chains fit together to form a complete protein. Quaternary protein structure is held together by hydrophobic interactions, and disulfide bridges.
What kind of interactions are not part of tertiary protein structure?
What kinds of interactions are NOT part of tertiary protein structure? peptide bonds.
What is the function of quaternary protein structure?
Functions of Quaternary Structure As mentioned above, quaternary structure allows a protein to have multiple functions. It also allows for a protein to undergo complicated conformational changes. This has several mechanisms. First, an individual subunit can change shape.
Which of the following does not show quaternary structure?
3. Which of the following does not possess a quaternary structure? Explanation: Myoglobin is a monomer, so it does not possess a quaternary structure.
What interactions occur in the secondary structure tertiary structure quaternary structure?
The secondary structure consists of local packing of polypeptide chain into α-helices and β-sheets due to hydrogen bonds between peptide bond – central carbon backbone. Tertiary (3D) structure is a shape resulting from folding of secondary structures determined by interactions between side chains of amino acids.
What bonds interactions hold primary structure together?
Hydrogen bonds. What bonds or interactions hold tertiary structure together? Hydrogen, ionic, disulfide, and hydrophobic. What bonds or interactions hold quaternary structure together?
What other interactions stabilize tertiary and quaternary structure?
The quaternary structure is also stabilized by the non-covalent interactions and disulfide bonds as in the tertiary structure, where more than one polypeptide is held together to form a single functional unit called multimer.
What stabilizes protein structure?
Hydrogen bonding in the polypeptide chain and between amino acid “R” groups helps to stabilize protein structure by holding the protein in the shape established by the hydrophobic interactions. … Interactions called van der Waals forces also assist in the stabilization of protein structure.
Which level of protein structure is disrupted through the hydrolysis of peptide bonds?
Which level of protein structure is disrupted through the hydrolysis of peptide bonds? The primary structure of a protein is the sequence of amino acids held together by peptide bonds. Peptide bonds are formed by dehydration reactions and disrupted by hydrolysis.
What does SDS do to lipids?
In the case of SHIELD and our technologies, the detergent Sodium Dodecyl Sulfate (SDS) is used to dissolve the lipids in the cell membranes to improve tissue transparency.
Does Sarkosyl denature proteins?
Sarkosyl is also a strong detergent and can denature many proteins, but leaves others unchanged.
Why does alcohol separate out DNA?
Since DNA is insoluble in ethanol and isopropanol, the addition of alcohol, followed by centrifugation, will cause the DNA proteins to come out of the solution. When DNA concentration in the sample is heavy, the addition of ethanol will cause a white precipitate to form immediately.
Does SDS-PAGE break disulfide bonds MCAT?
Since SDS is able to break down all non-covalent bonds within a protein and BME is able to break down all intermolecular and intramolecular disulfide bonds, SDS-PAGE under reducing conditions can be used to separate a completely denatured and isolated monomeric subunit of a peptide.