Atoms in Group 2A have 2 valence electron and lose 2 electron to gain an octet. These cations have +2 charge..
Beside this, what are Group 2a elements likely to do when they form ions gain electrons or lose them?
More specifically, they will always lose 2 electrons to form 2+ cations. In the case of beryllium, which is located in group 2, period 1, losing 2 electrons allows it to have a stable duet configuration, i.e. 2 electrons in its outermost shell, or valence shell.
Also Know, which groups lose or gain electrons? Elements that are metals tend to lose electrons and become positively charged ions called cations. Elements that are nonmetals tend to gain electrons and become negatively charged ions called anions. Metals that are located in column 1A of the periodic table form ions by losing one electron.
Beside this, do Group 2 elements gain or lose electrons?
Group 2 atoms lose two electrons to form positively charged ions. For example, magnesium atoms form Mg2+ ions. These have the same electron configuration as the noble gas neon. Most elements in Group 3 lose three electrons to form 3+ ions.
Does sodium gain or lose electrons?
The sodium atom wants to lose an electron and the chlorine atom wants to gain an electron. If you look at the diagram the sodium ion (a cation) now contains only ten electrons and the new chloride ion (an anion) has eighteen electrons. However, both ions now have eight electrons in their outer shell.
Related Question Answers
Do nonmetals gain electrons?
Metals: Metals like to lose valence electrons to form cations to have a fully stable octet. Nonmetals: Nonmetals like to gain electrons to form anions to have a fully stable octet. They release energy (exothermic) to gain electrons to form an anion; thus, electron affinity of nonmetals is higher than that of metals. ?What usually forms the positive ion?
An atom or a group of atoms that has an electric charge. Positive ions, or cations, are formed by the loss of electrons; negative ions, or anions, are formed by the gain of electrons.How do you tell if an element will gain or lose electrons?
Just determine whether it is easier to lose the electrons to become stable or gain them. If they have less than three valence electrons, it is easier to lose them and become a positively charged ion. If they have more than four valence electrons, it is easier to gain electrons and become negatively charged.What type of ions do group 2 elements form?
Explanation: Group 1 metals, the alkali metals, have the 1 valence electron, and thus form M+ ions when oxidized. Group 2 metals, the alkaline earth metals, have 2 valence electrons, and thus form M2+ ions. The halogens, Group 17 , reach a full valence shell upon reduction, and thus form X− ions.How many electrons does NA need to gain to lose a stable configuration?
Cations. A neutral sodium atom is likely to achieve an octet in its outermost shell by losing its one valence electron. The cation produced in this way, Na+, is called the sodium ion to distinguish it from the element. The outermost shell of the sodium ion is the second electron shell, which has eight electrons in it.What is the charge on the ions formed by Group 2a?
Atoms in Group 2A have 2 valence electron and lose 2 electron to gain an octet. These cations have +2 charge.Can all elements form ions?
An element's location on the periodic table can give a clue as to the type of ions the atoms of that element will form. Notice that all metals lose electrons to form positive ions. Group 1 metals commonly lose only one electron to form ions with a single positive charge.What makes an ionic compound?
Ionic compounds are compounds made up of ions. These ions are atoms that gain or lose electrons, giving them a net positive or negative charge. Metals tend to lose electrons, so they become cations and have a net positive charge. Nonmetals tend to gain electrons, forming anions that have a net negative charge.How many electrons does nitrogen gain or lose?
But typically a nitrogen atom gains 3 electrons to form the nitride ion, N3− .How many electrons does boron gain or lose?
2 Answers. Boron atomic number 5 has five electrons in its ground state. Commonly Boron will lose 3 electrons leaving 2 electrons in its most common ionic form.How many valence electrons does each group have?
Following this rule: Elements in group 1 have one valence electron; elements in group 2 have two valence electrons; elements in group 13 have three valence electrons; elements in group 14 have four valence electrons; and so forth up to group 18.Do halogens lose or gain electrons?
Metals tend to lose electrons and nonmetals tend to gain electrons. Halogens tend to gain one electron so that they can get a full outer level and be stable.Why do group 1 elements lose electrons?
Group 1 metals all have one electron in their outer shell. When they react they form positive metal ions by losing this electron. This means that the electrostatic forces of attraction between the outer shell electron and the nucleus are weaker and therefore it takes less energy for the electron to be lost.How do you determine valence electrons?
For neutral atoms, the number of valence electrons is equal to the atom's main group number. The main group number for an element can be found from its column on the periodic table. For example, carbon is in group 4 and has 4 valence electrons. Oxygen is in group 6 and has 6 valence electrons.Why do elements want to lose electrons?
Atoms that lose electrons acquire a positive charge as a result because they are left with fewer negatively charged electrons to balance the positive charges of the protons in the nucleus. Positively charged ions are called cations. Most metals become cations when they make ionic compounds.How many valence electrons does group 16 have?
six valence electrons
Why do group 2 elements form 2+ ions?
Like the alkali metals, the alkaline earth metals are so reactive that they are never found in elemental form in nature. Because they form +2 ions that have very negative reduction potentials, large amounts of energy are needed to isolate them from their ores.Which group gains electrons most easily?
The noble gases (group 18) elements are inert because of a full valence shell. Alkali Metals ?Group 1 metals on the periodic table that contain 1 valence electron and lose their valence electrons the most easily, making them the most reactive metals.How many electrons does strontium lose?
Strontium atom is a group 2, element so highly electropositive atom; therefore, they loss two electron to form ion. Hence, Strontium atom loss two electron so that they fulfil the octet rule or the noble gas electronic configuration. Strontium has 38 electrons. 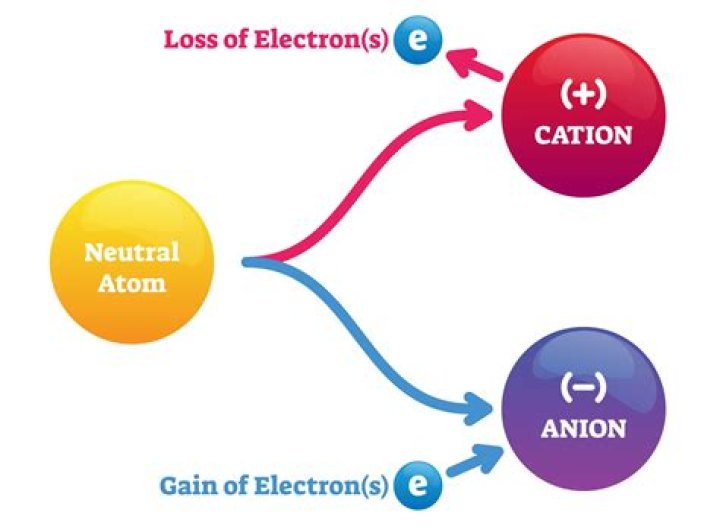
 Christopher Martinez
Christopher Martinez