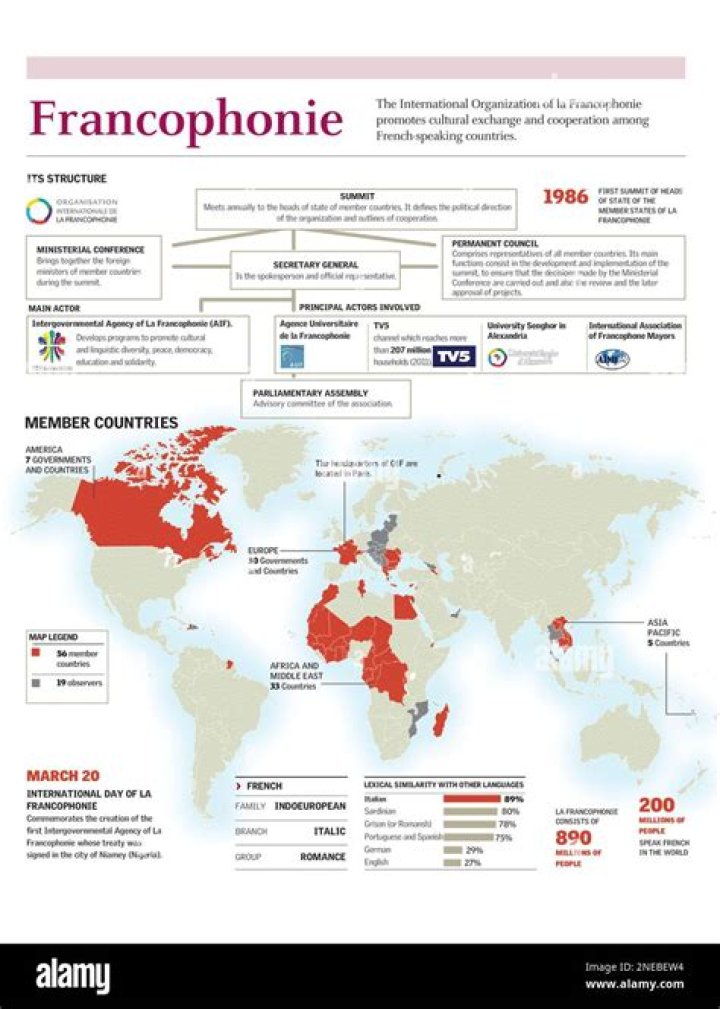
Does Boyles law apply to liquids
 Emma Terry
Emma Terry In contrast to gases, liquids are not compressible as their particles are already very close together. Boyle’s law only applies to gases.
Does Boyle's law only apply to gases?
Boyle’s Law holds true only if the number of molecules (n) and the temperature (T) are both constant. Boyle’s Law is used to predict the result of introducing a change in volume and pressure only, and only to the initial state of a fixed quantity of gas.
In which process Boyle's law is applicable?
Boyle’s law is applicable for an isothermal process, wherein temperature of gas remains constant.
Is Charles law applicable for liquids?
This should be a refresher for most engineers on the basic behavior of gases and liquids. Boyle’s, Charles’, and Pascal’s Laws describe the basic behavior of fluids with respect to volume, pressure, and temperature. … Pascal’s principle applies to a confined incompressible fluid.Which one is not Boyle's Law?
The graph C does not represent Boyle’s law as it is a straight line however the curve of P vs V for Boyle’s law is an exponential curve. The graph D indicates the equation P=−V+constant whereas the Boyles law is represented by the equation PV= constant. Explanation: graph C does not represent boyle’s law.
What are the assumptions of Boyle's Law?
The assumptions made in Boyle’s law are the constant temperature and number of moles of gases or amount of gas, the gas is within a closed system and lastly, the gas behaves ideally. An ideal gas is defined as a theoretical gas whose interactions are perfectly elastic collisions following the ideal gas law.
What is Boyle's gas law?
This empirical relation, formulated by the physicist Robert Boyle in 1662, states that the pressure (p) of a given quantity of gas varies inversely with its volume (v) at constant temperature; i.e., in equation form, pv = k, a constant. …
What is the difference between Boyle's Law and Charles Law?
In Charles law, temperature and volume of the gas are kept at constant pressure. Whereas in Boyle’s law, pressure and volume of the gas are kept at a constant temperature. In Boyle’s law, pressure and volume vary inversely whereas, in Charles law, pressure and volume vary directly.Is a hydraulic jack Boyle's Law?
The oil then applies pressure to a sleeve/piston that lifts the car. This is the same principle used in hydraulic jacks. … Mathematically, Boyle’s law can be written as pV=k, where p is the pressure of the gas, V is the volume of the gas, and k is a constant.
Why Boyles Law is applicable to ideal gases?Boyle’s law is good for an ideal gas. Real gases have interactions between atoms or molecules that are more than just kinetic collisions. There’s the van der Waals force, for instance, when the molecules get very close together. One can approximate an ideal gas by reducing its density sufficiently.
Article first time published onWhy Boyle's law is applicable to ideal gas?
A modern statement of Boyle’s law is: The absolute pressure exerted by a given mass of an ideal gas is inversely proportional to the volume it occupies if the temperature and amount of gas remain unchanged within a closed system. … Similarly, as volume decreases, the pressure of the gas increases.
Does adiabatic process obey Boyle's Law?
Answer: An ideal gas undergoes an adiabatic process obeying the relation PV^4/3 = constant.
What is incorrect according to Boyle's Law?
Boyle’s law states that P∝V1 or PV= constant. So, PV value shows a constant value but in Option (C) PV shows decreasing value. So, option (c) is incorrect.
Which of the following represents combination of Boyle's Law and Charles Law?
Combined gas law comes from the combination of Boyle’s law, Charles law and Gay Lussac’s law. According to Boyle’s law, At constant temperature, pressure of a gas is inversely proportional to its volume.
What is constant in Charles Law?
Charles’s Law states that the Volume (V) of a gas is directly proportional to the temperature (T). This law is valid as long as the pressure and the amount of gas are constant. The temperature must be an absolute temperature: VT=k(constant) The constant, k, will depend on the number of moles and the pressure.
How does Boyle's law apply in space?
Boyle’s law states that the pressure exerted by a gas (of a given mass, kept at a constant temperature) is inversely proportional to the volume occupied by it. The Boyle’s Law helps astronauts know how much of pressurized gas they can fit into a container without bursting and wasting any space.
What is an example of Boyle's Law?
An example of Boyle’s law in action can be seen in a balloon. Air is blown into the balloon; the pressure of that air pushes on the rubber, making the balloon expand. If one end of the balloon is squeezed, making the volume smaller, the pressure inside increased, making the un-squeezed part of the balloon expand out.
How did Boyle discover Boyle's Law?
Using Hooke’s pump, Boyle and Hooke carried out experiments to investigate the properties of air and the vacuum, making their first great discovery: Boyle’s Law. They made their discovery using a glass tube similar to the one shown at the top of this page.
What is an expression of Boyle's Law K constant?
Simply put, Boyle’s states that for a gas at constant temperature, pressure multiplied by volume is a constant value. The equation for this is PV = k, where k is a constant. At a constant temperature, if you increase the pressure of a gas, its volume decreases. If you increase its volume, the pressure decreases.
What relationship does Boyle's law describe apex?
Boyle’s Law is a relationship between pressure and volume. In this relationship, pressure and volume have an inverse relationship when temperature is held constant. If there is a decrease in the volume there is less space for molecules to move and therefore they collide more often, increasing the pressure.
How does temperature affect Boyle's Law?
Boyle’s Law – states that the volume of a given amount of gas held at constant temperature varies inversely with the applied pressure when the temperature and mass are constant. … If the amount of gas in a container is decreased, the volume decreases. This is assuming of course that the container has expandible walls.
Which law is applied in hydraulics?
PRESSURE AND FORCE IN HYDRAULIC SYSTEMS According to Pascal’s law, any force applied to a confined fluid is transmitted uniformly in all directions throughout the fluid regardless of the shape of the container.
What law is used in hydraulics?
Pascal’s law states that when there is an increase in pressure at any point in a confined fluid, there is an equal increase at every other point in the container.
What is Pascal's law hydraulics?
Pascal’s principle, also called Pascal’s law, in fluid (gas or liquid) mechanics, statement that, in a fluid at rest in a closed container, a pressure change in one part is transmitted without loss to every portion of the fluid and to the walls of the container.
Why is Boyle's law true?
According to Boyle’s Law, an inverse relationship exists between pressure and volume. Boyle’s Law holds true only if the number of molecules (n) and the temperature (T) are both constant.
Is Boyle's Law direct?
When the volume and the number of molecules remain constant. Charles Law is a direct relationship between temperature and volume. … The differences are that Boyle’s Law is a direct relationship while Charles Law is an inverse relationship. Both laws involve volume but one involves pressure and the other temperature.
What are the 4 gas laws?
Gas Laws: Boyle’s Law, Charle’s Law, Gay-Lussac’s Law, Avogadro’s Law.
How does Boyle's law relate to Charles Law?
Explanation: Boyle’s Law tells us that for an ideal gas, at a constant temperature, volume is proportional to pressure. … Charles’ Law tells us that for an ideal gas, at constant pressure, volume is proportional to temperature (where temperature is in degrees Kelvin).
Why are liquids instead of gases used in hydraulic brakes?
Liquids are used in hydraulic systems because they are more difficult to compress than gases, so are better at transmitting forces – when a force is applied at one end of the system, it is easy transferred through the system.
What is Boyle's Law and Charles Law in chemistry?
Boyle’s law—named for Robert Boyle—states that, at constant temperature, the pressure P of a gas varies inversely with its volume V, or PV = k, where k is a constant. … Charles (1746–1823)—states that, at constant pressure, the volume V of a gas is directly proportional to its absolute (Kelvin) temperature T, or V/T = k.
Why Boyle's law is not applicable for non ideal gases?
Particles of a hypothetical ideal gas have no significant volume and do not attract or repel each other. … The gas therefore becomes less compressible at these high pressures, and although its volume continues to decrease with increasing pressure, this decrease is not proportional as predicted by Boyle’s law.