Does actin have a quaternary structure
 Isabella Bartlett
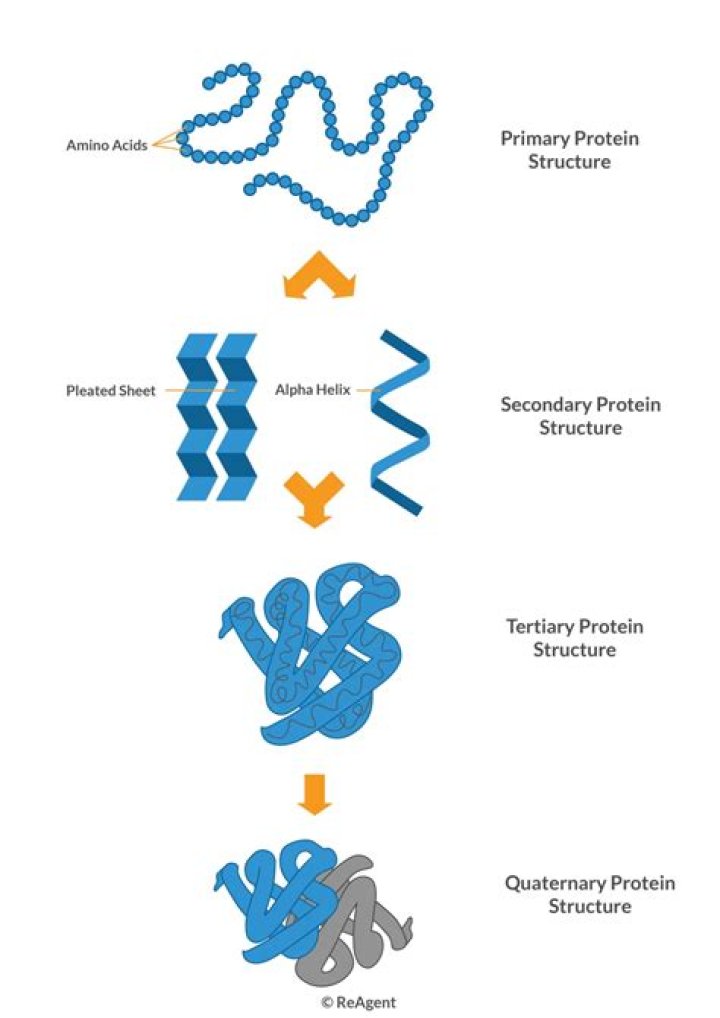
Isabella Bartlett Quaternary structure refers to the way in which the subunits of such proteins are assembled in the finished protein. … These proteins exist in a soluble globular form that can assemble into long helical filaments called microfilaments (actin) and microtubules (tubulin) (Figure 21 ).
What is the structure of actin?
The actin structure is highly conserved. The actin monomer consists of two major domains each of which contains two subdomains. The four subdomains are organized to form a rather flat molecule. Two large clefts are formed between the two major domains of actin.
What type of protein structure is actin?
Actin is a family of globular multi-functional proteins that form microfilaments in the cytoskeleton, and the thin filaments in muscle fibrils. It is found in essentially all eukaryotic cells, where it may be present at a concentration of over 100 μM; its mass is roughly 42 kDa, with a diameter of 4 to 7 nm.
Which protein has a quaternary structure?
Hemoglobin has a quaternary structure. It consists of two pairs of different proteins, designated the α and β chains.Is actin a tertiary structure?
Each F-actin monomeric unit has, as part of its tertiary structure, several loops that are important to its assembly to the polymeric F-actin.
Is actin a structural protein?
Actin belongs to a structural superfamily with sugar kinases, hexokinases, and Hsp70 proteins (3). The Arp proteins (49) and the prokaryotic actin-like proteins MreB (54) and ParM (56) are also now part of this superfamily.
What is actin in muscle contraction?
Actin and myosin filaments work together to generate force. This force produces the muscle cell contractions that facilitate the movement of the muscles and, therefore, of body structures.
Do all proteins have quaternary structure?
In contrast to the first three levels of protein structure, not all proteins will have a quaternary structure since some proteins function as single units. Protein quaternary structure can also refer to biomolecular complexes of proteins with nucleic acids and other cofactors.Is albumin a quaternary structure?
Human serum albumin complexed with dodecanoic acid, pdb id 1E7F, is the most common protein found in blood plasma. … With a primary sequence length of 585 amino acids and a quaternary structure composed of a single subunit, human serum albumin, or HSA, is a relatively small, heart-shaped protein (6, 5).
How do you know if a protein has a quaternary structure?The quaternary structure (QS) of a protein is determined by measuring its molecular weight in solution. The data have to be extracted from the literature, and they may be missing even for proteins that have a crystal structure reported in the Protein Data Bank (PDB).
Article first time published onIs actin a dimer?
Filamin (also called actin-binding protein or ABP-280) binds actin as a dimer of two 280-kd subunits. The actin-binding domains and dimerization domains are at opposite ends of each subunit, so the filamin dimer is a flexible V-shaped molecule with actin-binding domains at the ends of each arm.
Is actin found in connective tissue?
The cells of all connective tissues (except blood) contain structural proteins in the form of fibres. … Actin is a myoprotein that is found in muscle cells and helps in the contraction of muscles.
Is actin a carbohydrate lipid or protein?
actin, protein that is an important contributor to the contractile property of muscle and other cells. It exists in two forms: G-actin (monomeric globular actin) and F-actin (polymeric fibrous actin), the form involved in muscle contraction.
Why does actin Treadmilling occur?
Treadmilling is a phenomenon observed in many cellular cytoskeletal filaments, especially in actin filaments and microtubules. It occurs when one end of a filament grows in length while the other end shrinks resulting in a section of filament seemingly “moving” across a stratum or the cytosol.
Is actin a transport protein?
In addition to the intracellular transport of particles (cargo) along microtubules, there are in the cell two actin-based transport systems. … Obligatory components of the actin-based transport are proteins of the WASP/Scar family and a complex of Arp2/3 proteins.
Is actin a motor protein?
Myosin is an actin motor protein, where myosin serves as the engine, the actin filaments provide the tracks that myosin can move along and the energy source that fuels the movement is adenosine triphosphate (ATP).
Is actin involved in mitosis?
In higher plants, microtubule (MT)-based, and actin filament (AF)-based structures play important roles in mitosis and cytokinesis.
How does actin help with mitosis?
The network of actin filaments is one of the crucial cytoskeletal structures contributing to the morphological framework of a cell and which participates in the dynamic regulation of cellular functions. … This actin cytoskeleton is reorganized during mitosis to form rounded cells with increased cortical rigidity.
Whats the definition of actin?
Definition of actin (Entry 1 of 2) : a cellular protein found especially in microfilaments (such as those comprising myofibrils) and active in muscular contraction, cellular movement, and maintenance of cell shape. actin- combining form.
What type of protein is actin and myosin?
In summary, myosin is a motor protein most notably involved in muscle contraction. Actin is a spherical protein that forms filaments, which are involved in muscle contraction and other important cellular processes. Tropomyosin is a long strand that loops around the actin chains in the thin filament.
What does the actin protein do?
Actin is a highly abundant intracellular protein present in all eukaryotic cells and has a pivotal role in muscle contraction as well as in cell movements. Actin also has an essential function in maintaining and controlling cell shape and architecture.
Are actin and myosin fibrous proteins?
Myosin combines easily with another muscle protein called actin, the molecular weight of which is about 50,000; it forms 12 to 15 percent of the muscle proteins. Actin can exist in two forms—one, G-actin, is globular; the other, F-actin, is fibrous.
Is albumin tertiary structure?
X-ray crystallography has shown albumin to possess a heart-shaped tertiary structure, but in solution HSA is ellipsoid. … HSA contains 35 cysteine residues, most of which form disulfide bridges (17 in all), contributing to overall tertiary structure.
What determines quaternary structure?
Quaternary Structure: Protein Chains Combine to Make Protein Complexes. Secondary and tertiary structures are determined by a protein’s sequence of amino acids, or primary structure. … Some proteins are made up of more than one amino acid chain, giving them a quaternary structure.
What is the structure of albumin?
Human albumin is a small globular protein (molecular weight: 66.5 kDa), consisting of a single chain of 585 amino acids organized in three repeated homolog domains (sites I, II, and III). Each domain comprises two separate sub-domains (A and B) 5.
Which proteins do not have quaternary structure?
Hemoglobin, p53 and DNA polymerase are all composed of subunits, while myoglobin is a functional single sequence. Since myoglobin does not have multiple subunits, it does not have quaternary structure.
Why all proteins have quaternary structure?
Many proteins are made up of a single polypeptide chain and have only three levels of structure (the ones we’ve just discussed). However, some proteins are made up of multiple polypeptide chains, also known as subunits. When these subunits come together, they give the protein its quaternary structure.
Are all enzymes quaternary?
Explanation: It would completely depend on the enzyme we’re considering. Some are only monomeric (e.g. trypsin), some contain several subunits which interact to form a quaternary structure. … In the case of trypsin, a serine protease, we could say the enzyme only has a tertiary structure.
Is a monomer a quaternary structure?
Quaternary structure may be defined as the specific association of subunits to form a single functional molecule. The arrangement of the subunits (monomers) and the way they pack together is of great importance since small changes in their relative orientations can cause marked changes in the properties of the protein.
Why don t all proteins form a quaternary structure?
All proteins have primary, secondary and tertiary structures but quaternary structures only arise when a protein is made up of two or more polypeptide chains. The folding of proteins is also driven and reinforced by the formation of many bonds between different parts of the chain.
Which of the following are an essential part of quaternary structure?
Quaternary structure is stabilized primarily by hydrophobic interactions. Quaternary structure exists only in proteins containing more than one polypeptide. Quaternary structure is fine-tuned by ion pairs, disulfide bonds, and hydrogen bonds.